Med Spa Infection Control and Clinical Safety Standards
Med spa infection control is a systematic approach to preventing healthcare-associated infections and cross-contamination within aesthetic treatment environments. These protocols include OSHA-aligned bloodborne pathogen training, high-level disinfection of treatment rooms, and the use of sterile, single-use supplies to ensure a safe clinical environment for both practitioners and clients.
Regulatory Compliance and Safety Standards
Every med spa must operate under a strict clinical safety framework that aligns with state boards and federal OSHA regulations. This foundation protects the license of the facility and the health of the community.
Compliance begins with a written infection control plan that is reviewed annually by the clinical director. This document outlines specific steps for handling sharps, managing blood exposure, and maintaining hygiene logs.
Staff training is a mandatory component of a safe facility. All clinical employees must complete bloodborne pathogen training to recognize and mitigate risks associated with injectable treatments and skin resurfacing.
Treatment Room Sanitation Systems
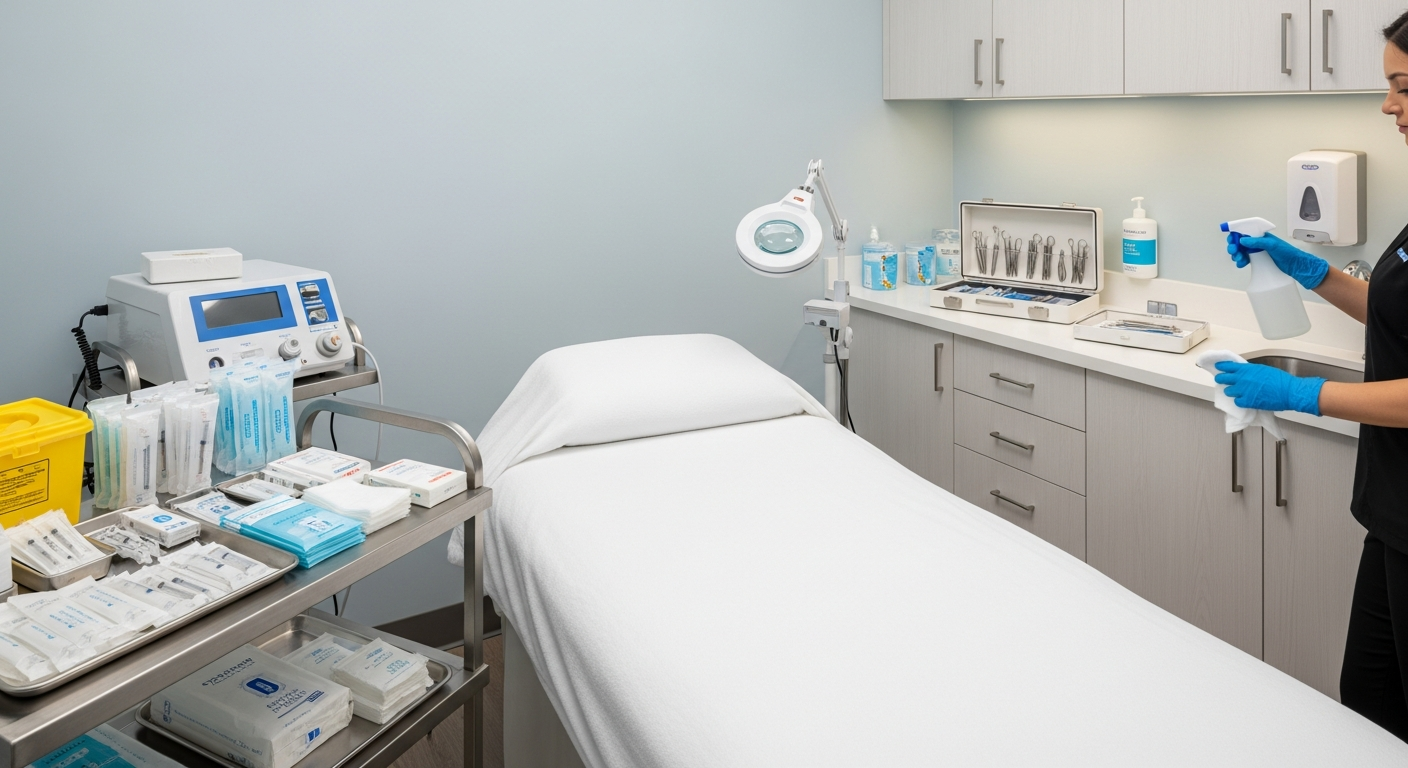
Pre-Procedure Room Preparation
Before any client enters the treatment room, all high-touch surfaces must be cleaned and disinfected with EPA-registered solutions. This includes treatment tables, counters, and magnifying lamps.
Barriers should be used on equipment that is difficult to clean thoroughly. Protective sleeves or films provide an additional layer of safety between clients and electronic devices.
The practitioner must perform a thorough hand-washing sequence using antimicrobial soap before donning new, medical-grade gloves. Hand hygiene is the most effective way to stop the spread of pathogens.
Post-Procedure Decontamination
After a treatment concludes, all single-use items like gauze, needles, and applicators must be disposed of immediately. Never leave contaminated items on a tray or counter.
Surfaces must stay wet with disinfectant for the full contact time recommended by the manufacturer to ensure total microbial kill. Rushing this process can lead to ineffective sanitation.
The treatment table must be wiped down entirely, even if protective paper was used. Fabric or porous surfaces should be covered with non-porous barriers to allow for proper chemical disinfection.
Tool and Device Decontamination
Reusable instruments must undergo a multi-step sterilization process. This begins with manual cleaning to remove organic debris, followed by high-level disinfection or autoclaving.
Steam sterilization is the gold standard for any tool that penetrates the skin or contact mucous membranes. Autoclaves must be monitored weekly with biological indicators to verify they are functioning correctly.
Stored instruments must remain in sealed, dated sterilization pouches. If a pouch is punctured or damp, the contents are no longer considered sterile and must be reprocessed.
Preventing Cross-Contamination in Procedures
Cross-contamination often occurs when a practitioner touches a non-sterile surface and then touches the client or sterile supplies. Proper “clean field” techniques are essential for every procedure.
Avoid reaching into multi-use containers with contaminated gloves. Use sterile spatulas or tongue depressors to dispense products like numbing creams or ultrasound gel into secondary containers.
Labels and expiration dates must be checked regularly. Using expired products or compromised packaging increases the risk of introducing bacteria into the treatment area.
PPE Requirements for Clinical Staff
Personal Protective Equipment (PPE) acts as a barrier between the practitioner and potential infectious materials. This includes gloves, masks, and occasionally eye protection or gowns.
Gloves must be changed between every client and whenever they become torn or heavily soiled. PPE should never be worn outside of the treatment room or into common areas.
Masks are necessary during procedures that involve close proximity to the client’s face or when using equipment that may create plumes or splashes of fluids.
Biohazardous Waste Management
Proper disposal of sharps is a critical med spa infection control practice. Sharps containers must be puncture-resistant, leak-proof, and located at the point of use.
Red biohazard bags should be used for waste that is saturated with blood or other potentially infectious materials. These bags must be stored in a secure, designated area until pickup.
A certified medical waste hauler should be contracted to remove biohazardous materials from the med spa. Manifests and disposal records must be kept for compliance audits.