Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic implementation of sanitation, disinfection, and sterilization protocols designed to prevent the spread of pathogens during aesthetic treatments. These OSHA-aligned practices protect both clients and practitioners by establishing rigorous barriers against cross-contamination through proper hand hygiene, surface disinfection, and sterile tool management.
Core Sanitation Practices for Treatment Rooms
Every treatment room must undergo a thorough disinfection process between every client.
Practitioners must use EPA-registered disinfectants on all high-touch surfaces and equipment.
This includes treatment tables, counters, stools, and any digital device interfaces.
All porous items or single-use materials must be discarded immediately after use.
Linens must be changed and laundered using high-temperature cycles and clinical-grade detergents.
Maintaining a clutter-free environment reduces the surface area available for environmental contaminants.
Surface Disinfection and Room Turnover
- Pre-cleaning: Removing visible debris before applying disinfectant solutions.
- Contact Time: Ensuring surfaces remain wet with disinfectant for the manufacturer-specified duration.
- Barrier Protection: Using disposable covers on equipment that cannot be easily saturated.
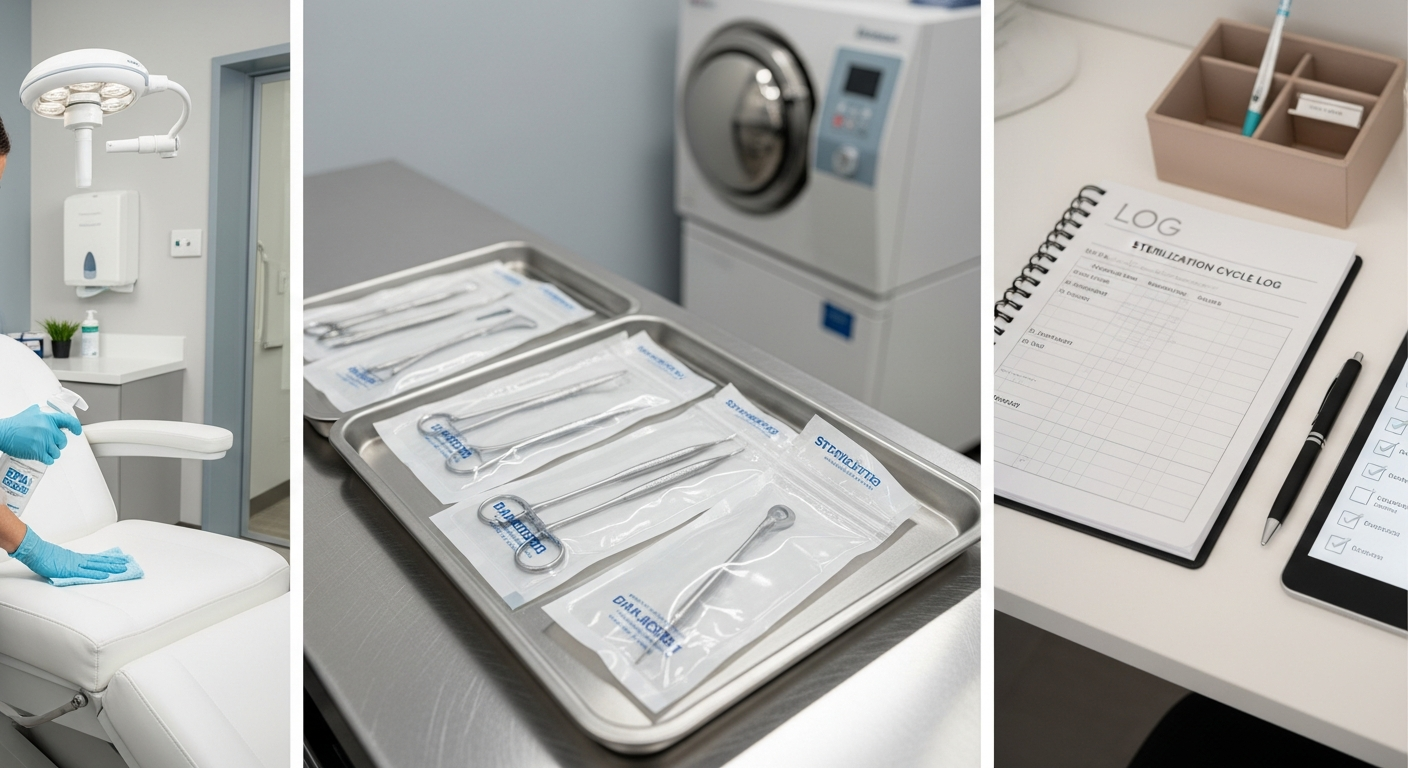
Sterilization Standards for Clinical Tools
Any instrument that penetrates the skin or contacts mucosal membranes requires high-level sterilization.
Med spas must use an autoclave to ensure all biological pathogens are completely neutralized.
Chemical cold sterilization is only appropriate for tools that cannot withstand high heat.
Sterilized tools must remain in sealed, dated pouches until the exact moment of use.
Practitioners should check sterilization indicators to confirm the process reached necessary parameters.
Proper storage in clean, closed cabinets prevents re-contamination of processed instruments.
Instrument Processing and Storage
- Decontamination: Cleaning tools in a dedicated area before the sterilization cycle.
- Indicator Tapes: Utilizing internal and external indicators to verify heat exposure.
- Biological Monitoring: Performing weekly spore tests to ensure autoclave functionality.
Preventing Cross-Contamination During Procedures
Personal protective equipment serves as a primary barrier against infectious pathogens.
Practitioners must change gloves between every stage of a clinical procedure to ensure safety.
Hands must be washed or sanitized before donning and after removing any protective gear.
Med spas should implement a “no-touch” policy for clean supplies during active treatments.
Using sterile tweezers to retrieve items from drawers prevents the contamination of bulk stock.
Proper waste segregation ensures that sharps and biohazardous materials are handled safely.
PPE and Hand Hygiene Protocols
- Glove Integrity: Replacing gloves immediately if they become punctured or heavily soiled.
- Facial Protection: Wearing masks and eye protection during any splash-prone aesthetic treatment.
- Aseptic Technique: Maintaining a sterile field when preparing needles or clinical devices.
Maintaining OSHA Compliance and Safety Logs
Written protocols ensure every team member follows the same infection control standards.
Med spas must maintain updated safety data sheets for every chemical used in the facility.
Regular training sessions keep the clinical staff informed about evolving sanitation requirements.
Documentation is a critical component of a professional clinical safety program.
Facilities must log every sterilization cycle, spore test result, and room cleaning schedule.
These records provide evidence of compliance during inspections and ensure operational consistency.
Documentation and Training Requirements
- Safety Logs: Maintaining daily records of equipment maintenance and cleaning tasks.
- Staff Training: Providing annual bloodborne pathogen training for all clinical employees.
- Waste Management: Following strict state guidelines for the disposal of medical waste.