Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent the spread of pathogens during aesthetic treatments. These practices ensure compliance with OSHA standards and protect both clients and practitioners by maintaining a sterile environment through rigorous treatment room management and cross-contamination prevention strategies.
OSHA-Aligned Sanitation and Personal Hygiene
Establishing a foundation of personal hygiene is the first step in med spa infection control.
Practitioners must follow strict hand-washing protocols using antimicrobial soap before and after
every client contact to eliminate transient microorganisms.
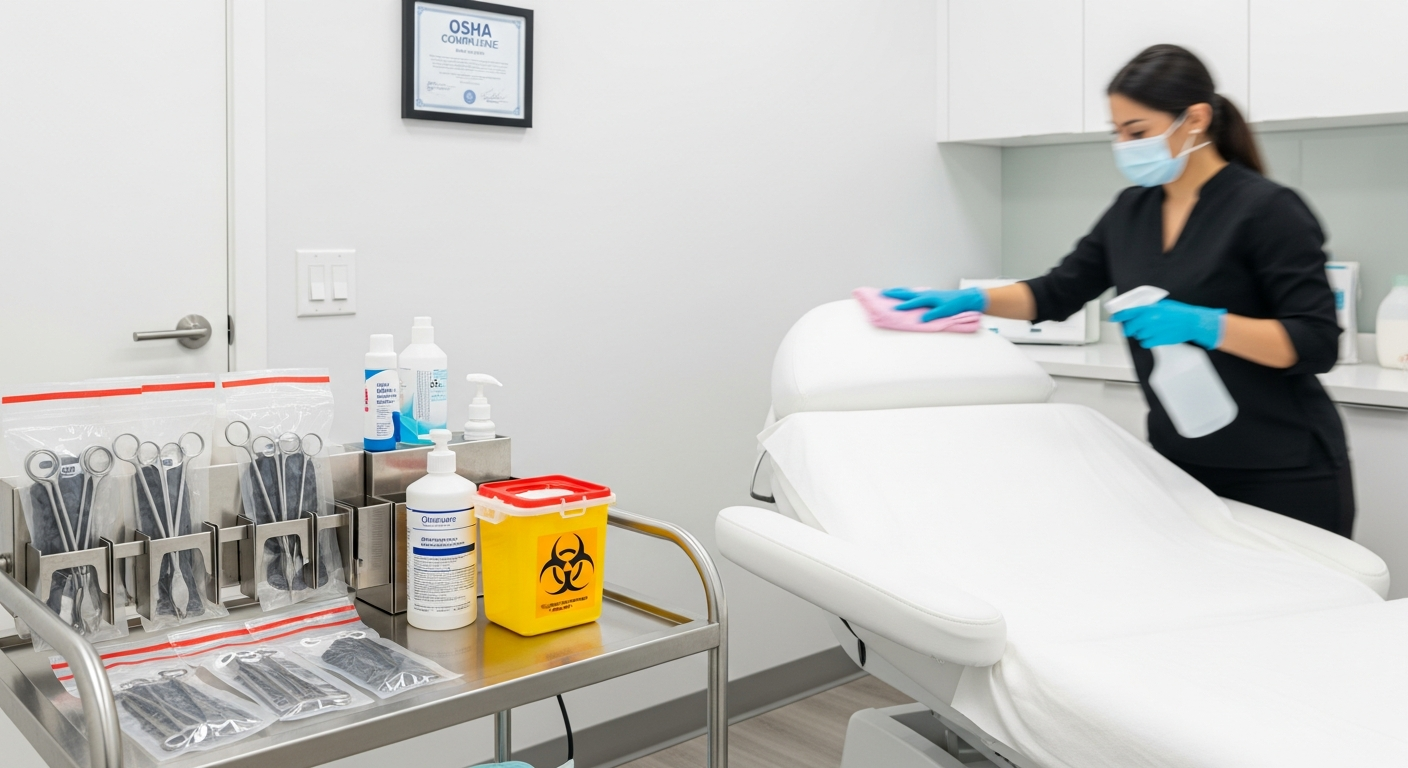
Personal Protective Equipment (PPE) serves as a critical barrier in clinical settings.
Med spa staff must utilize medical-grade gloves, masks, and eye protection
wherever there is a risk of exposure to bloodborne pathogens or bodily fluids.
Compliance managers should ensure that all staff members are trained on OSHA standards.
This includes the proper donning and doffing of PPE and the immediate
disposal of contaminated materials in designated biohazard containers.
Treatment Room Disinfection and Surface Protocols
Every treatment room requires a systematic disinfection process between appointments.
All non-porous surfaces, including treatment tables, stools, and work counters,
must be wiped down with EPA-registered, hospital-grade disinfectants.
Contact time is a vital component of effective room turnover.
Disinfectants must remain wet on surfaces for the full duration specified
by the manufacturer to ensure the complete neutralization of viruses and bacteria.
High-touch areas such as door handles, equipment buttons, and magnifying lamps
require frequent cleaning throughout the day. Using disposable barrier films
on difficult-to-clean equipment further enhances the facility’s safety profile.
Sterilization Processes for Clinical Implements
Any reusable tool that penetrates the skin or contacts mucosal membranes
requires high-level sterilization. Med spas should utilize autoclaves
that use steam under pressure to achieve total microbial destruction.
Strict monitoring of the sterilization cycle is necessary for compliance.
Clinical supervisors must use chemical indicators and perform weekly biological
spore tests to verify that the equipment is functioning according to safety standards.
Sterilized implements must remain in sealed, dated pouches until the moment of use.
If a pouch is punctured or damp, the tools inside are considered
contaminated and must undergo the entire sterilization process again.
Preventing Cross-Contamination in Aesthetic Settings
Cross-contamination prevention relies on the clear separation of clean and soiled items.
Med spas should implement a one-way flow system where contaminated tools
never enter the same space as sterilized equipment or clean supplies.
Single-use items must be discarded immediately after a single application.
Items such as needles, lancets, and certain applicators should never be
reused or cleaned, and sharps must be placed in puncture-resistant containers.
-
Core Prevention Strategies:
- Use of disposable barriers on electronic equipment.
- Maintaining dedicated “clean” and “dirty” utility zones.
- Implementing no-touch waste receptacles in all treatment areas.
- Regular auditing of clinical logs and sanitation schedules.
By maintaining these systematic safety processes, med spa owners protect their
reputation and legal standing. Consistent adherence to clinical protocols
ensures a safe environment for both the aesthetic team and the clientele.