Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent the spread of pathogens within aesthetic environments. These practices include OSHA-aligned bloodborne pathogen training, treatment room decontamination, and the use of medical-grade barriers to ensure patient and provider safety during cosmetic procedures.
Standard Precautions in Aesthetic Settings
Every med spa must operate under the principle of standard precautions. This means treating all biological materials as potentially infectious to protect staff and clients.
Consistent hand hygiene is the foundation of clinical safety. Providers must wash hands thoroughly before and after every patient contact and after removing gloves.
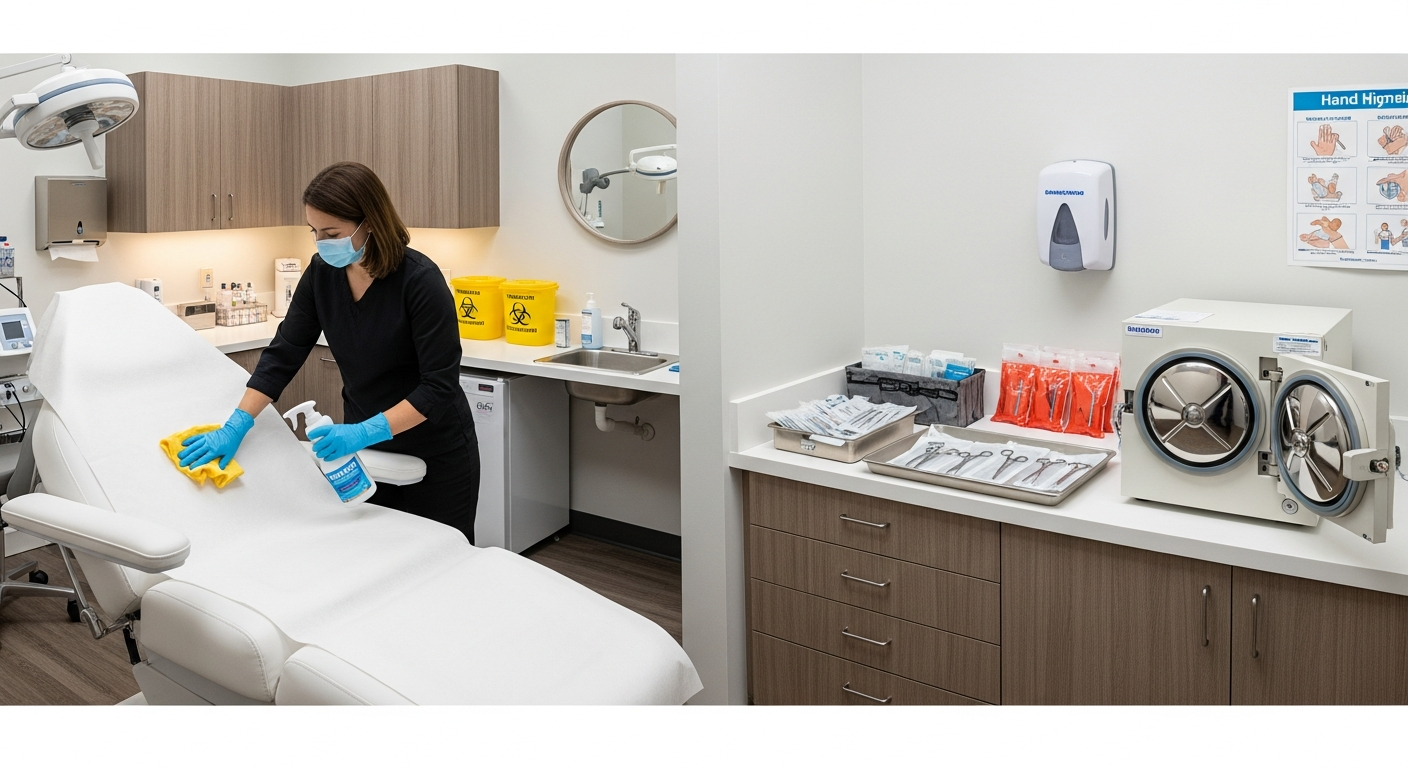
Personal Protective Equipment (PPE) serves as a vital physical barrier. This typically includes single-use gloves, face masks, and eye protection during treatments involving fluids.
Treatment Room Disinfection Systems
Treatment rooms require rigorous cleaning between every patient appointment. All contact surfaces must be wiped down with EPA-registered, intermediate-level disinfectants.
Surface Decontamination Steps
- Remove all used linens and disposable barriers immediately after the procedure.
- Apply disinfectant to countertops, treatment tables, and equipment handles.
- Ensure the disinfectant remains wet on the surface for the full required contact time.
Protective barriers, such as plastic sheathing or medical-grade paper, should be used on high-touch areas. These are replaced between every session to prevent cross-contamination.
Sterilization Standards for Aesthetic Tools
Any tool that penetrates the skin or contacts compromised tissue must undergo full sterilization. Multi-use metal implements require mechanical processing in an autoclave.
Proper sterilization involves pre-cleaning to remove visible debris before placing items in sterilization pouches. Pouches must feature chemical indicators to verify heat exposure.
Biological monitoring, or spore testing, should be conducted weekly. This ensures the autoclave is successfully destroying the most resistant forms of microbial life.
High-Level Disinfection for Non-Critical Items
Items that only touch intact skin are considered non-critical. These require high-level disinfection rather than full sterilization in most clinical jurisdictions.
Providers must distinguish between porous and non-porous materials. Porous items like sponges or wooden applicators are strictly single-use and must be discarded.
Cross-Contamination Prevention Strategies
Preventing the transfer of bacteria requires a strict “clean to dirty” workflow. This organizational strategy keeps sterile supplies separate from used instruments.
Avoid “double-dipping” into bulk product containers during treatments. Use sterile spatulas to transfer products into secondary, disposable containers before starting the procedure.
Waste management is equally critical for safety. All sharps must be placed in puncture-resistant containers, and biohazardous waste must be bagged and labeled correctly.
OSHA-Aligned Compliance and Training
U.S. med spas must adhere to OSHA Bloodborne Pathogens Standards. This includes maintaining an Exposure Control Plan that is updated and reviewed annually.
Staff training should cover the correct handling of chemicals and emergency protocols. Safety Data Sheets (SDS) for all disinfectants must be accessible to all employees.
Regular safety audits help identify gaps in sanitation protocols. Documenting cleaning schedules and sterilization logs creates an accountable culture of clinical excellence.