Med Spa Infection Control and Clinical Safety
Med spa infection control is a set of clinical protocols used to prevent the transmission of infectious agents during aesthetic treatments. These practices include OSHA-aligned hand hygiene, high-level disinfection of surfaces, and the sterilization of multi-use instruments. Establishing these systematic safety processes ensures a sterile environment and reduces risks of cross-contamination for all clients.
OSHA-Aligned Sanitation and Hygiene Practices
Sanitation is the foundation of a safe med spa environment. Every facility must follow Occupational Safety and Health Administration guidelines to protect staff and clients.
This includes maintaining accessible handwashing stations equipped with liquid soap and disposable towels. Providers must perform hand hygiene before and after every client contact.
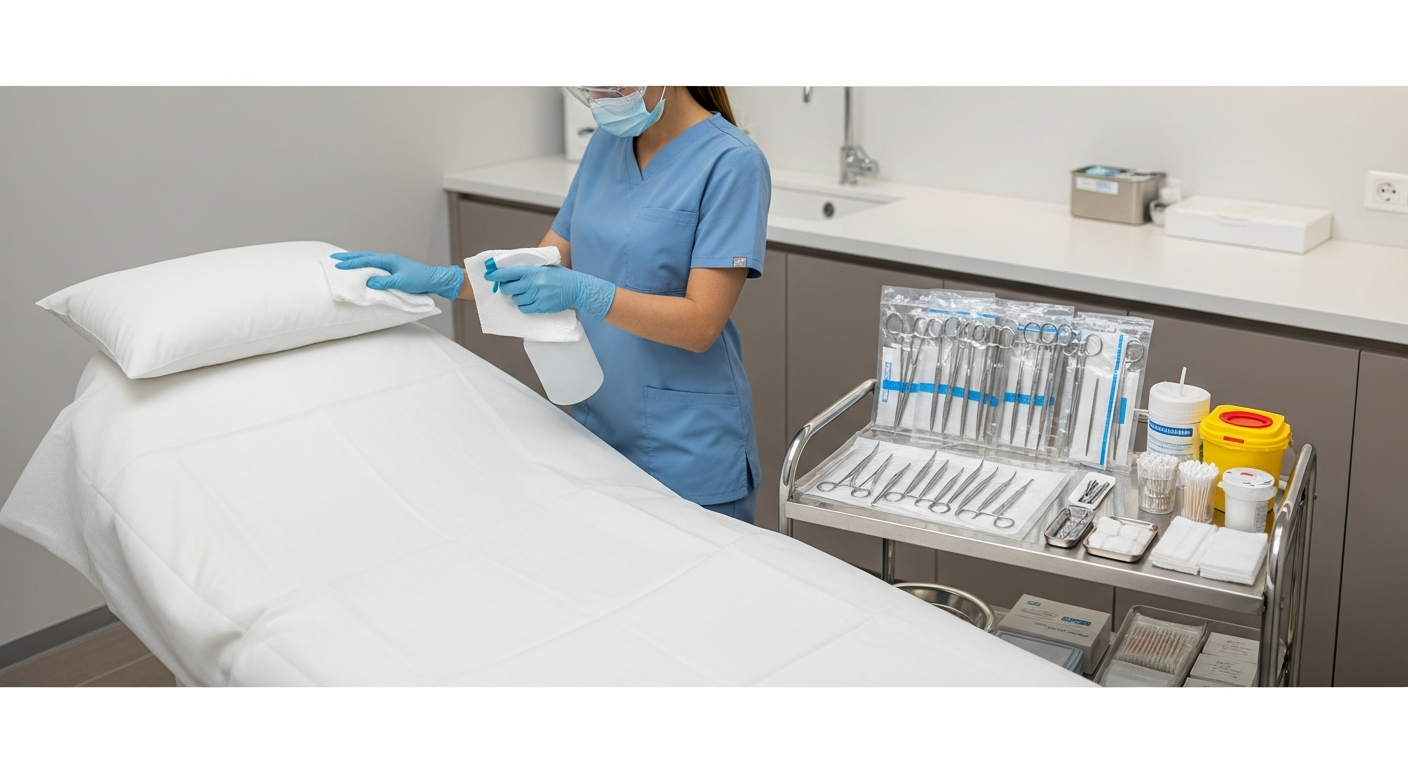
Hand Hygiene and Protective Barriers
Hand hygiene remains the most effective way to prevent the spread of pathogens. Alcohol-based rubs are used when hands are not visibly soiled during treatment transitions.
Personal Protective Equipment acts as a vital barrier between the provider and the client. Gloves, masks, and eye protection must be used based on the specific risk of the procedure.
Gloves must be changed between every client and never reused. Proper donning and doffing techniques prevent the accidental spread of contaminants to clean surfaces.
Sterilization Standards for Clinical Instruments
The med spa must distinguish between items that require simple cleaning and those that require high-level sterilization. Any tool that penetrates the skin must be sterile.
Reusable metal tools must undergo a multi-stage decontamination process. This involves manual cleaning to remove debris followed by mechanical sterilization in an autoclave.
Decontamination and Autoclave Protocols
Cleaning is the essential first step before sterilization can occur. Instruments must be scrubbed with enzymatic cleaners to ensure the sterilization process is effective.
Autoclaves use pressurized steam to kill all microbial life, including bacterial spores. Regular biological monitoring and chemical indicators verify that the equipment is functioning correctly.
Sterilized items must remain in sealed pouches until the moment of use. If a pouch is punctured or damp, the instrument is no longer considered safe for clinical use.
Treatment Room Disinfection Systems
Surface disinfection is a critical component of med spa infection control. High-touch surfaces like treatment tables, counters, and stools require constant attention.
EPA-registered disinfectants must be used according to the manufacturer’s specified contact time. This “wet time” ensures that pathogens are actually neutralized on the surface.
Treatment tables should be covered with disposable barriers that are replaced after every session. This adds an extra layer of protection against skin-to-surface contact.
Cross-Contamination Prevention Strategies
Cross-contamination occurs when pathogens are transferred from one surface or person to another. Rigid operational systems are necessary to block these pathways.
Med spa staff must maintain a clear distinction between “clean” and “dirty” areas. Instruments should never move from a contaminated zone back into a sterile storage area.
Single-Use Item Management
The safest way to prevent infection is the use of disposable, single-use items. This includes needles, lancets, gauze, and certain applicators used during aesthetic procedures.
All sharps must be disposed of immediately in puncture-resistant containers. These containers must be located as close as possible to the point of use to minimize transport risk.
Soft porous materials cannot be sterilized and must be discarded after a single use. This practice ensures that no organic material is carried over between different client appointments.