Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the rigorous implementation of sanitation, disinfection, and sterilization standards to prevent the transmission of infectious agents in aesthetic clinics. These protocols ensure compliance with OSHA regulations by establishing systematic barriers against cross-contamination during facial, injectable, and laser-based treatments.
Core Sanitation and OSHA-Aligned Practices
Aesthetic facilities must maintain a clean environment to protect both practitioners and clients.
OSHA compliance requires a written exposure control plan that outlines specific steps for
handling bloodborne pathogens and other potentially infectious materials.
Daily operations should focus on reducing bioburden through systematic cleaning of all high-touch
surfaces. This includes reception areas, consultation desks, and staff break rooms to ensure
pathogens do not migrate into clinical zones.
Safety training is a critical component of med spa infection control.
Staff must be educated on the proper dilution of chemical disinfectants and the
correct use of safety data sheets for all cleaning agents used on-site.
Clinical Disinfection in Treatment Rooms
Every treatment room functions as a controlled clinical environment.
Between every client appointment, all non-porous surfaces must undergo a
thorough decontamination process using EPA-registered disinfectants.
Surface Decontamination Protocols
Practitioners must wipe down treatment tables, stools, and equipment carts
after each procedure. Electronic devices and laser consoles should be
protected with disposable barriers or cleaned with compatible clinical wipes.
Linens used during treatments must be changed after every client.
Soiled laundry should be handled with gloves and processed using high-temperature
wash cycles and professional-grade detergents to ensure complete pathogen removal.
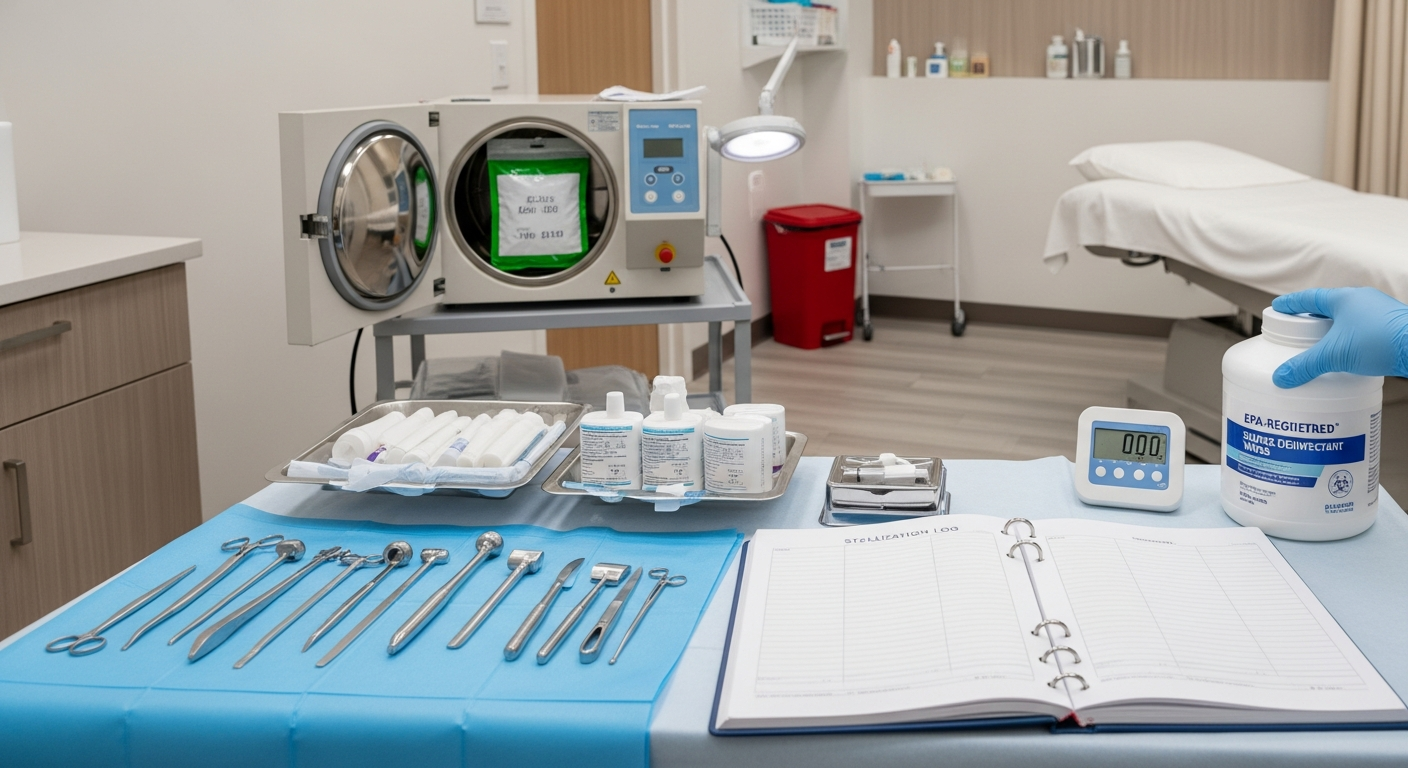
Sterilization and Instrument Maintenance
Any instrument that penetrates the skin or comes into contact with bodily fluids
requires high-level sterilization. Med spas should utilize autoclaves that
use saturated steam under pressure to destroy all microbial life.
- Biological Monitoring: Perform weekly spore tests to verify autoclave efficacy.
- Chemical Indicators: Use internal and external indicators on every sterilized pouch.
- Storage: Keep sterilized tools in sealed, dated pouches until the moment of use.
- Single-Use Items: Dispose of needles, lancets, and wooden applicators immediately.
Maintaining a detailed sterilization log is essential for clinical accountability.
Each entry should record the date, time, cycle temperature, and the
initials of the staff member responsible for the sterilization load.
Preventing Cross-Contamination in Aesthetic Settings
Cross-contamination often occurs through improper hand hygiene or the
misuse of personal protective equipment. Establishing strict zones for
clean and dirty items helps minimize the risk of spreading contaminants.
Personal Protective Equipment and Hand Hygiene
Hands must be washed with antimicrobial soap before and after every client
contact. Alcohol-based hand rubs are effective for intermediate steps,
but physical washing is required when hands are visibly soiled.
Gloves must be changed between different tasks on the same client to
prevent transferring bacteria from one area to another. Protective eyewear
and masks should be worn during procedures that generate splashes or sprays.
Waste management is the final step in the infection control chain.
Biohazardous waste, such as used needles and blood-soaked gauze, must
be placed in puncture-resistant, labeled containers for professional disposal.