Med Spa Infection Control and Clinical Safety
Med spa infection control is the implementation of rigorous sanitation, disinfection, and sterilization protocols designed to eliminate the transmission of pathogens in aesthetic environments. These practices include following OSHA bloodborne pathogen standards, utilizing medical-grade personal protective equipment, and maintaining strict treatment room hygiene to protect both practitioners and clients during clinical procedures.
Core Principles of Med Spa Infection Control
Infection control in a med spa setting relies on the concept of universal precautions. This approach requires staff to treat all human blood and bodily fluids as if they are known to be infectious.
Establishing a culture of safety involves continuous training on pathogen transmission. Clinical supervisors must ensure every team member understands how to break the chain of infection during treatments.
OSHA-Aligned Sanitation and Hygiene Standards
Compliance with OSHA standards is mandatory for maintaining a safe clinical environment. This includes having a written exposure control plan that is updated annually to reflect new safety technologies.
Hand hygiene serves as the primary defense against cross-contamination. Practitioners must perform thorough hand washing with antimicrobial soap before and after every client contact or glove change.
Personal Protective Equipment Protocols
The use of personal protective equipment (PPE) is essential during aesthetic procedures. This includes medical-grade gloves, masks, and eye protection to prevent exposure to splashes or spray.
Staff must receive training on the proper sequence for donning and doffing PPE. This prevents the accidental transfer of contaminants from used equipment to clean skin or surfaces.
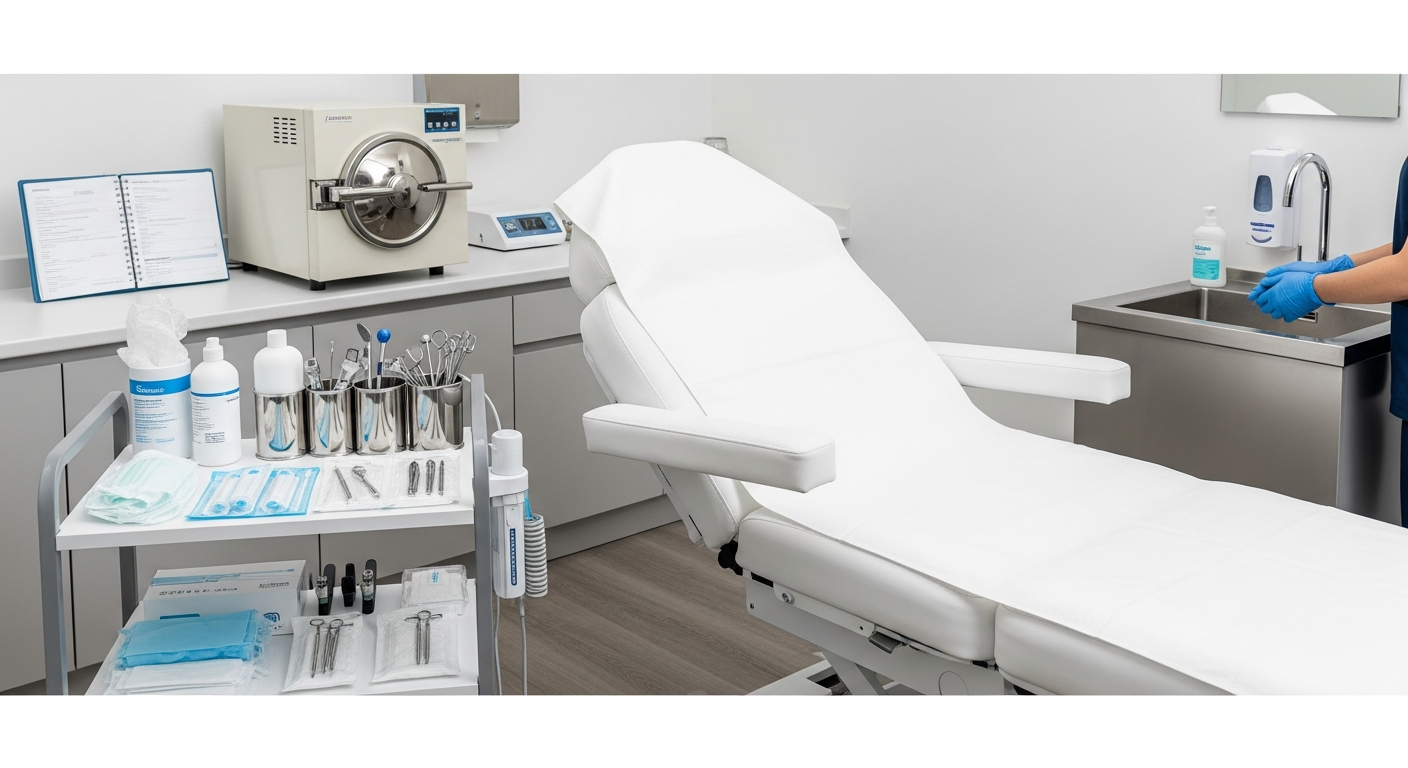
Treatment Room Disinfection Systems
Treatment room surfaces must be disinfected between every client visit. This applies to all high-touch areas, including treatment tables, equipment carts, and adjustable stools.
Only EPA-registered, hospital-grade disinfectants should be used for surface cleaning. These agents must remain wet on the surface for the full contact time specified by the manufacturer.
Environmental Cleaning and Air Quality
Regular cleaning schedules must extend beyond the immediate treatment area. Floors, cabinetry, and air filtration systems require routine maintenance to minimize the presence of environmental dust and microbes.
Sterilization Standards for Aesthetic Tools
Any instrument that penetrates the skin or contacts compromised tissue must undergo full sterilization. This process ensures the complete destruction of all microbial life, including bacterial spores.
Autoclaves are the standard for sterilization in the med spa industry. These machines use pressurized steam to reach the temperatures necessary for total decontamination of reusable metal tools.
Sterilization Monitoring and Documentation
Facilities must implement a system for verifying that the sterilization process was successful. This involves using chemical indicators on every load and biological indicators on a weekly basis.
Detailed logs must be maintained for every sterilization cycle. These records should include the date, time, temperature, pressure, and the initials of the technician who processed the items.
Cross-Contamination Prevention Systems
Preventing cross-contamination requires a logical workflow that separates clean supplies from used materials. Dedicated “clean” and “dirty” zones help prevent the accidental reuse of contaminated items.
Single-use items, such as needles and disposable applicators, must be discarded immediately after use. Sharps must be placed in puncture-resistant, leak-proof containers located at the point of use.
Standardizing these safety processes ensures that every treatment is performed under optimal clinical conditions. Consistent adherence to these protocols protects the health of the community and the facility.