Med Spa Infection Control and Clinical Safety Protocols
Med spa infection control is the systematic implementation of sanitation and sterilization protocols designed to eliminate the transmission of infectious agents during aesthetic treatments. These practices include the use of EPA-registered disinfectants, strict adherence to OSHA standards, and the maintenance of a sterile environment to protect both practitioners and clients.
Foundational OSHA-Aligned Sanitation Practices
Adhering to federal safety standards ensures that every treatment area meets the baseline requirements for clinical safety.
Med spas must implement a written exposure control plan to manage bloodborne pathogens and other potential contaminants.
This plan provides a roadmap for staff to follow during routine cleaning and emergency spills.
Standard and Universal Precautions
Universal precautions dictate that all human blood and certain body fluids are treated as if known to be infectious.
Practitioners must treat every client interaction with the same high level of scrutiny regarding fluid contact.
This mindset minimizes the risk of accidental exposure during invasive or minimally invasive procedures.
Personal Protective Equipment Usage
The consistent use of personal protective equipment, such as medical-grade gloves and masks, is a core safety requirement.
Gloves must be changed between every client and after any action that compromises their integrity.
Appropriate eye protection should be worn whenever there is a risk of splashing or aerosolization of fluids.
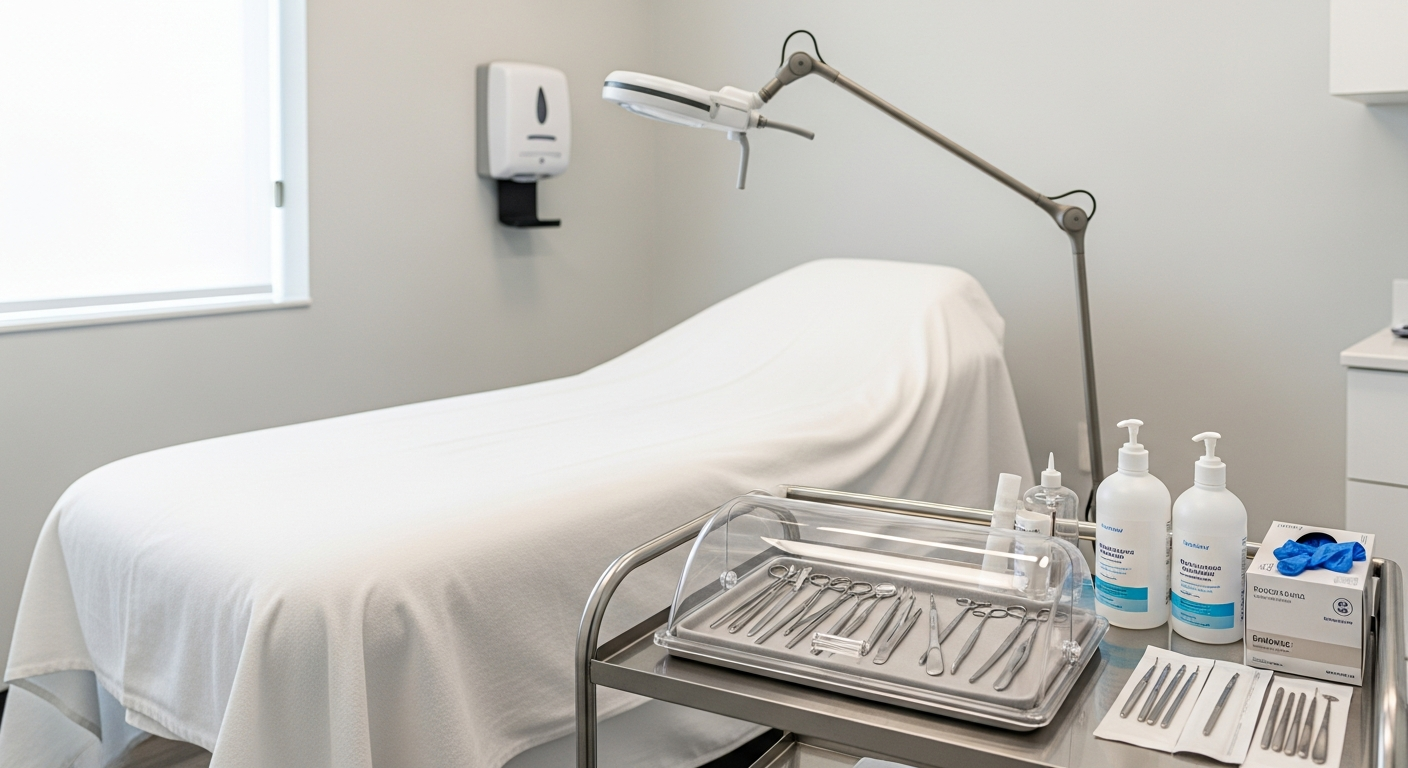
Treatment Room Disinfection Systems
Treatment rooms require a tiered approach to cleaning that distinguishes between general surfaces and high-risk contact areas.
Every surface must be non-porous to allow for effective chemical decontamination between scheduled appointments.
A systematic checklist ensures that no area is overlooked during the transition from one client to the next.
Environmental Surface Cleaning
Clinical surfaces such as treatment tables, counters, and stools must be cleaned with intermediate-level disinfectants.
The disinfectant must remain wet on the surface for the full contact time specified by the manufacturer to be effective.
Failure to observe these dwell times can result in incomplete pathogen eradication.
High-Touch Point Decontamination
High-touch points like doorknobs, light switches, and equipment control panels require frequent disinfection throughout the day.
These areas are primary vectors for cross-contamination between different treatment rooms and common areas.
Using disposable barrier films on electronic controls provides an additional layer of protection for sensitive equipment.
Sterilization and Cross-Contamination Prevention
Effective med spa infection control relies on the strict separation of clean and contaminated items within the facility.
Clearly labeled zones help staff identify where used tools end and sterile supplies begin.
Maintaining this physical boundary is essential for preventing the migration of microbes through the clinic.
Instrument Processing Standards
Any reusable tool that penetrates the skin or contacts mucous membranes must undergo professional-grade sterilization.
This process involves thorough pre-cleaning, followed by treatment in a validated autoclave or dry heat sterilizer.
Chemical indicators and periodic biological monitoring must be used to verify that the sterilization equipment is functioning correctly.
Single-Use Item Management
Single-use items such as needles, lancets, and certain applicators must be disposed of immediately after a single application.
Sharps must be placed in puncture-resistant, leak-proof containers that are labeled with biohazard symbols.
Proper disposal prevents accidental needle sticks and ensures that contaminated waste is handled by professional biohazard services.