Med Spa Infection Control: Essential Safety Protocols
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent the transmission of pathogens during aesthetic procedures. These clinical safety measures include OSHA-aligned bloodborne pathogen training, high-level disinfection of treatment surfaces, and the use of single-use disposables to eliminate cross-contamination risks for clients and staff.
OSHA-Aligned Sanitation Standards for Med Spas
Med spa facilities must adhere to strict sanitation standards to maintain a safe environment. This involves implementing universal precautions that assume all bodily fluids are potentially infectious.
Regulatory compliance requires annual training on bloodborne pathogen exposure for all clinical staff. Med spas must maintain updated exposure control plans and provide accessible safety data sheets for all cleaning agents.
Establishing a culture of safety begins with documented hand hygiene protocols. Staff should perform handwashing with antimicrobial soap before and after every client contact, regardless of glove use.
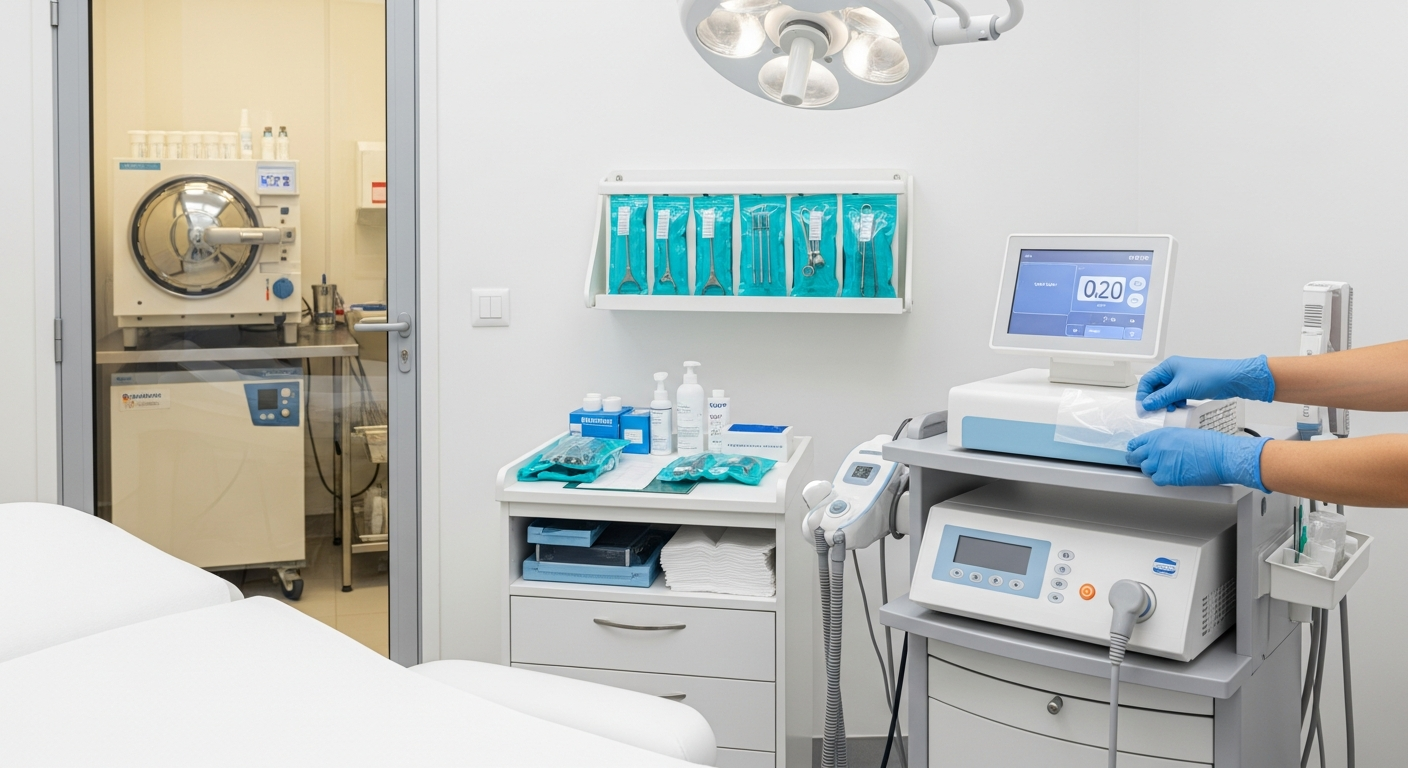
Treatment Room Disinfection Systems
Effective disinfection systems require a structured workflow between client appointments. All non-porous surfaces must be treated with EPA-registered disinfectants according to the manufacturer’s specified contact time.
Focus on high-touch areas such as treatment tables, adjustable stools, and equipment consoles. Using barrier films on electronic controls provides an additional layer of protection against surface contamination.
Linens and table covers must be changed after every session. Soiled laundry should be handled as biohazardous material and processed using high-temperature cycles to ensure complete decontamination.
Sterilization and Reusable Tool Management
Reusable instruments must undergo a multi-step sterilization process involving ultrasonic cleaning. This removes debris before tools enter a pressurized steam autoclave for final processing.
Med spas should monitor sterilization cycles with chemical indicators and periodic biological spore testing. Detailed logs must be maintained to track every load and ensure equipment is functioning correctly.
Sterilized tools should remain in sealed pouches until the point of use. If a pouch is punctured or damp, the contents must be considered contaminated and re-processed immediately.
Cross-Contamination Prevention Strategies
Eliminating cross-contamination relies on strict movement protocols and the separation of clean and soiled items. Technicians must avoid touching communal containers with contaminated gloves during treatments.
Single-use items like needles, lancets, and wooden applicators must be disposed of immediately after use. Sharps containers should be puncture-resistant, labeled correctly, and replaced when they reach the fill line.
Proper PPE selection is critical for procedure-specific safety. This includes utilizing single-use gloves, face masks, and eye protection whenever there is a risk of splashing or aerosolization.
- Daily: Disinfect all common areas and treatment workstations.
- Weekly: Perform biological monitoring of sterilization equipment.
- Monthly: Review and update safety compliance documentation.
Personal Protective Equipment Usage
PPE serves as the primary barrier between the practitioner and potential pathogens. Med spa owners must ensure all staff are fitted with appropriate sizes to maintain effective protection.
Gloves must be changed between every client and whenever they become torn or heavily soiled. Practitioners should never wash or reuse disposable gloves under any circumstances.