Med Spa Infection Control Protocols and Standards
Med spa infection control is a set of clinical protocols designed to eliminate the transmission of infectious agents during aesthetic procedures. These standards include systematic hand hygiene, the use of personal protective equipment, and the rigorous disinfection of environmental surfaces and medical instruments to maintain a sterile treatment environment.
OSHA-Aligned Sanitation Practices
Med spas must adhere to federal safety guidelines to protect both practitioners and clients from bloodborne pathogens. Standard precautions assume all body fluids are potentially infectious.
Hand hygiene serves as the foundation of every clinical safety program. Practitioners must wash hands with antimicrobial soap before and after every patient contact.
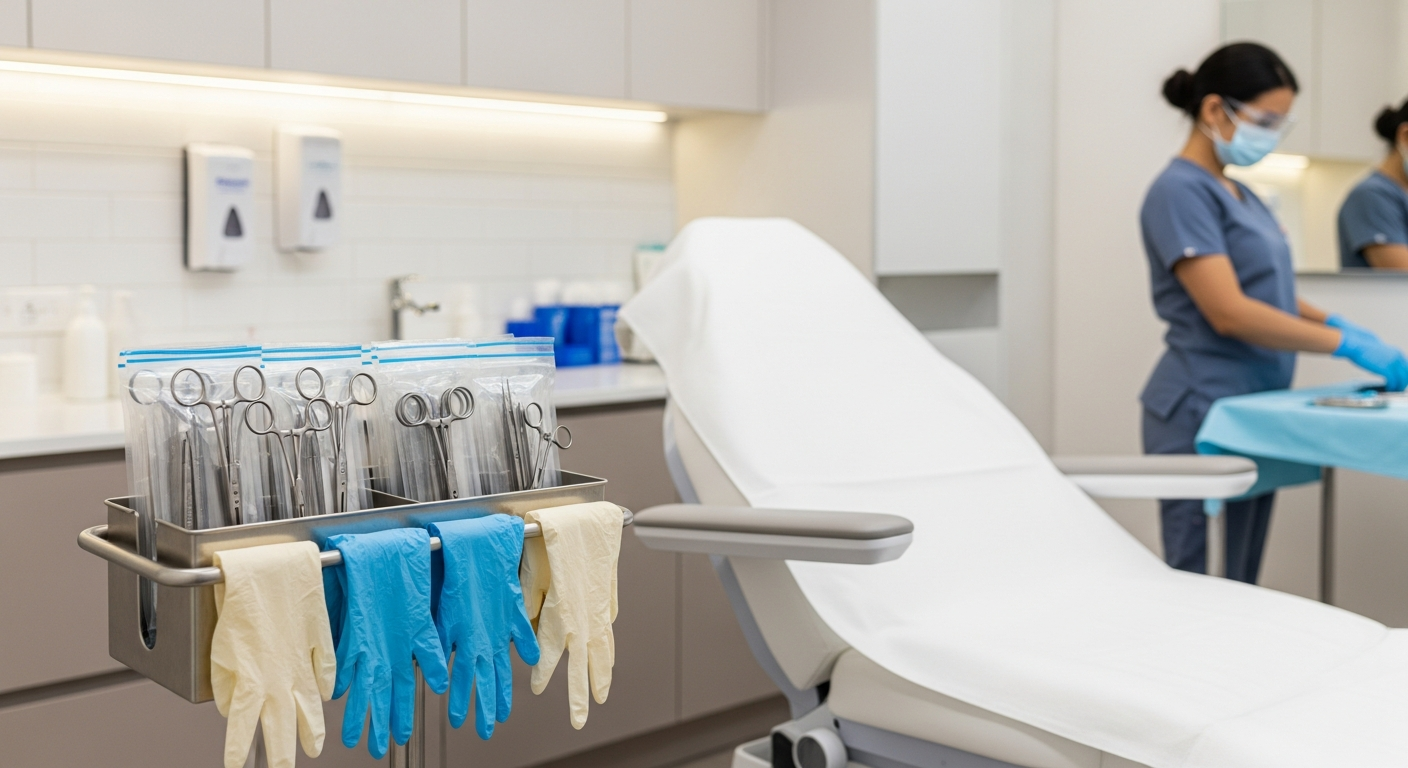
Personal Protective Equipment (PPE) provides a critical barrier against cross-contamination. This includes the consistent use of medical-grade gloves, masks, and eye protection during treatments.
Treatment Room Disinfection Systems
Treatment surfaces must be decontaminated between every client session using EPA-registered disinfectants. These solutions must be labeled for use in clinical settings.
Practitioners must follow the specific contact time listed on disinfectant labels. Surfaces must remain visibly wet for the full duration to ensure complete pathogen eradication.
High-touch areas require focused attention during the turnover process. This includes treatment tables, Mayo stands, stool handles, and digital device screens used during procedures.
Clinical Sterilization Standards
Any reusable instrument that penetrates the skin or contacts mucosal membranes requires high-level sterilization. Steam sterilization via autoclave is the gold standard for these tools.
Sterilization logs must track every cycle to ensure the equipment reaches necessary pressure and temperature. Biological indicators should be used weekly to verify the autoclave functions correctly.
Instruments must be cleaned of all organic debris before being placed in sterilization pouches. Pouches must remain sealed until the point of use in a sterile field.
Cross-Contamination Prevention
Single-use items must be disposed of immediately after use in appropriate containers. Needles and blades must go into puncture-resistant sharps containers to prevent injury.
Aseptic technique is required when handling injectable products or topical serums. This prevents the transfer of microorganisms from the environment into the product or the client.
Workstations should be organized to separate clean supplies from contaminated items. Establishing a one-way flow of materials minimizes the risk of accidental exposure during busy shifts.
Biohazardous Waste Management
Clinical waste containing blood or other potentially infectious materials must be handled separately from standard trash. Regulated waste requires specific labeling and specialized disposal services.
Staff training on waste segregation ensures that biohazard bins are not overfilled. This reduces the risk of environmental contamination during the removal and transport process.
Spill kits must be readily available in all treatment areas. Employees should be trained on the specific steps for neutralizing and cleaning biological spills safely.