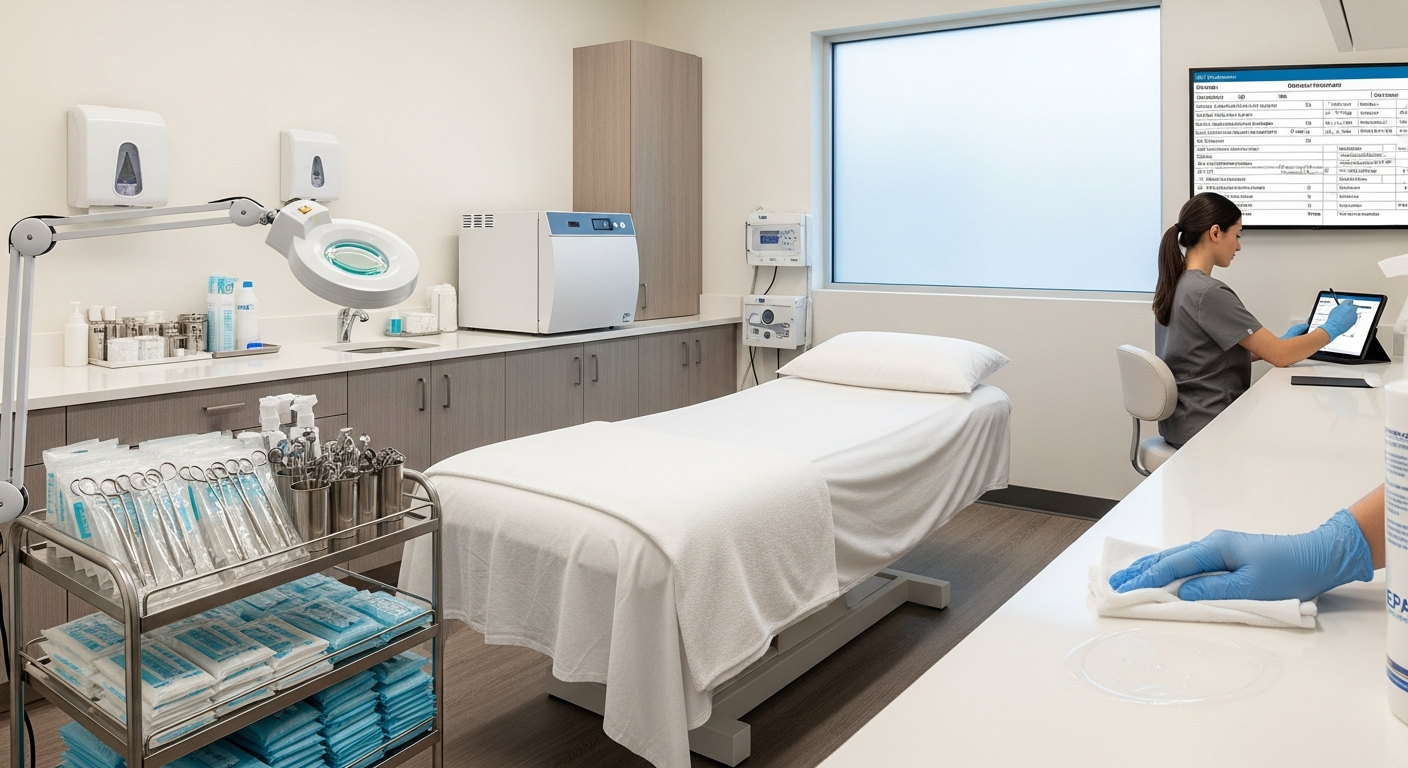
Med Spa Infection Control and Clinical Safety Standards
Med spa infection control is a systematic approach to maintaining clinical safety through rigorous sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination. These practices align with OSHA standards to protect clients and staff during aesthetic treatments by ensuring all tools, surfaces, and equipment are free from pathogenic microorganisms.
OSHA-Aligned Sanitation and Hygiene
Every med spa must implement a comprehensive hand hygiene program as the first line of defense against infection. Staff should use antimicrobial soap or alcohol-based rubs before and after every client contact.
Personal Protective Equipment (PPE) is mandatory for practitioners performing invasive or semi-invasive treatments. This includes the use of medical-grade gloves, masks, and eye protection to prevent fluid exposure.
Standard precautions assume that all blood and body fluids are potentially infectious. Clinical supervisors must ensure all staff are trained in handling biohazardous materials according to federal safety guidelines.
Treatment Room Disinfection Systems
Treatment rooms require a tiered approach to cleaning based on the level of contact with the client. Surface disinfection must occur between every appointment using EPA-registered, hospital-grade disinfectants.
High-Touch Surface Protocols
Focus on high-touch areas such as treatment tables, equipment buttons, and magnifying lamps. These surfaces must remain wet with disinfectant for the full contact time specified by the manufacturer.
Countertops and workstation surfaces should be non-porous to prevent the absorption of liquids or contaminants. Any surface that cannot be thoroughly disinfected must be covered with a single-use barrier.
Sterilization Standards for Aesthetic Tools
Any instrument that penetrates the skin or contacts mucosal membranes must undergo full sterilization. The industry standard is the use of a steam autoclave which utilizes heat and pressure.
Tools must be cleaned of all organic debris before being placed in sterilization pouches. Each pouch should feature a chemical indicator that changes color to confirm successful processing.
Clinical managers must maintain a sterilization log to track every cycle. This log should include the date, time, load number, and the results of biological monitoring tests.
Preventing Cross-Contamination in Med Spas
Cross-contamination is frequently caused by the improper handling of multi-use containers or the reuse of single-use items. All disposables, such as needles and lancets, must go into sharps containers.
Products like numbing creams or gels should be dispensed into a secondary container using a clean spatula. Never “double-dip” or allow the original container to come into contact with the treatment area.
Establishing a “clean to dirty” workflow prevents the spread of pathogens. This involves organizing the treatment room so that sterile supplies are physically separated from contaminated waste and used tools.
Monitoring and Compliance Systems
A successful safety program requires regular audits of infection control practices. Compliance managers should perform unannounced spot checks to ensure all sanitation steps are followed consistently.
Standard Operating Procedures (SOPs) must be printed and accessible to all clinical staff. These documents should outline the exact steps for room turnover and instrument processing in the med spa.
Ongoing education is essential for maintaining a safe clinical environment. Annual training refreshers on bloodborne pathogens and infection prevention help mitigate risks and ensure long-term operational safety.