Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic implementation of sanitation, disinfection, and sterilization protocols designed to prevent the spread of pathogens during aesthetic treatments. These OSHA-aligned practices include rigorous surface decontamination, proper hand hygiene, and the use of personal protective equipment to maintain a sterile environment for both clients and practitioners.
OSHA-Aligned Sanitation and Hygiene Practices
Every med spa must establish a comprehensive hygiene plan that adheres to OSHA Bloodborne Pathogens Standards. This involves mandatory handwashing protocols before and after every patient contact or glove change.
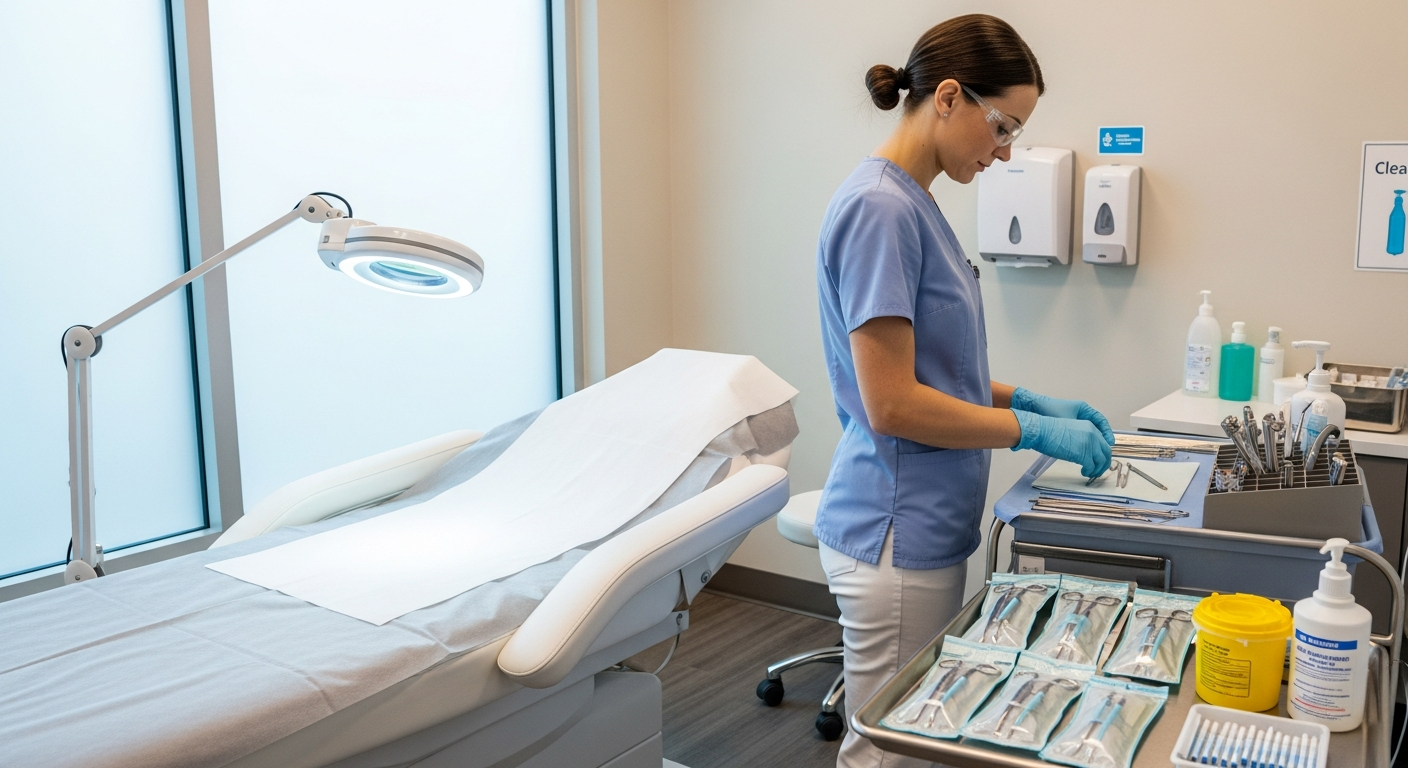
Practitioners must use appropriate personal protective equipment, including medical-grade gloves, masks, and eye protection. These barriers prevent the transmission of fluids and microorganisms during invasive or aerosol-generating procedures.
Safety data sheets for all cleaning agents should be easily accessible to staff. Training programs must be conducted annually to ensure every team member understands the correct handling of biohazardous materials.
Systematic Treatment Room Disinfection
Treatment rooms require a tiered approach to decontamination between every patient visit. All clinical surfaces, including treatment tables and counters, must be cleaned of visible debris before applying disinfectants.
High-Touch Surface Decontamination
Focus disinfection efforts on high-touch areas such as adjustable stool levers, magnification lamps, and product bottles. Use EPA-registered, intermediate-level disinfectants that are effective against a broad spectrum of pathogens.
Ensure that disinfectants remain wet on surfaces for the full contact time specified by the manufacturer. This dwell time is critical for the chemical to successfully neutralize bacteria, viruses, and fungi.
Clinical Sterilization Standards for Equipment
Any instrument that penetrates the skin or contacts mucosal membranes is considered a critical item. These tools must undergo a multi-stage cleaning and sterilization process rather than simple disinfection.
Autoclave Protocols and Monitoring
Steam sterilization via autoclave is the standard for reusable metal instruments in a med spa. Instruments must be cleaned of organic matter, dried, and bagged in indicator pouches before the cycle begins.
Clinical supervisors must perform regular biological monitoring, such as spore testing, to verify autoclave efficacy. Detailed logs should be maintained for every sterilization cycle to ensure full traceability and compliance.
Cross-Contamination Prevention Systems
Preventing cross-contamination requires strict zone management within the med spa facility. Clearly define “clean” and “dirty” areas to prevent used instruments from contacting sterile supplies.
Single-use disposables, such as needle tips and gauze, should be discarded immediately into designated sharps containers or biohazard bags. Never reach into drawers or supply cabinets with contaminated gloves during a procedure.
Implement a “no double-dipping” policy for topical products by using sterile spatulas to transfer creams to secondary containers. This ensures the original product reservoir remains uncontaminated for the next clinical use.