Med Spa Infection Control and Clinical Safety Standards
Med spa infection control involves a systematic set of sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination in aesthetic clinical environments. These OSHA-aligned practices include rigorous treatment room disinfection, proper use of personal protective equipment, and the maintenance of sterile tool environments to ensure client and practitioner safety during cosmetic procedures.
Standard Precautions for Aesthetic Treatments
Effective infection control begins with hand hygiene and the consistent use of personal protective equipment (PPE) during every procedure.
Practitioners must wash hands with antimicrobial soap before and after every client contact to reduce microbial transmission.
Gloves, masks, and eye protection serve as critical barriers against bloodborne pathogens and chemical exposure.
PPE should be changed between every client session to prevent carrying contaminants from one treatment area to another.
Disposable gloves must never be washed or reused, and hands should be sanitized immediately after glove removal.
Proper donning and doffing techniques ensure that the practitioner’s skin never contacts potentially infectious materials.
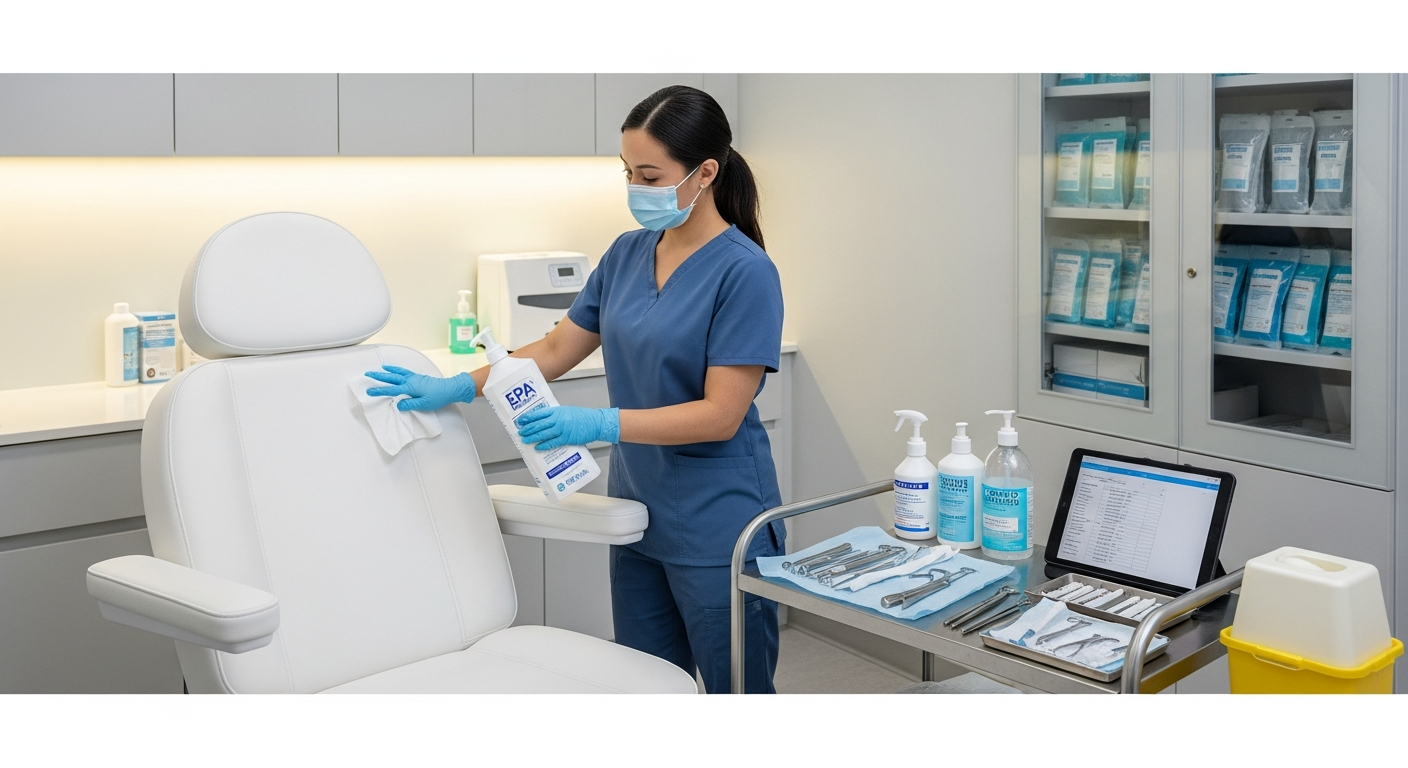
Treatment Room Disinfection Systems
Every treatment room requires a multi-step disinfection process between clients to maintain a safe clinical environment.
All high-touch surfaces, including treatment tables, stools, and product counters, must be wiped with EPA-registered disinfectants.
These chemicals must remain wet on the surface for the full contact time specified by the manufacturer.
Surface Sanitation Protocols
- Pre-Cleaning: Remove visible debris from surfaces using detergent before applying clinical disinfectants.
- Disinfection: Apply medical-grade wipes or sprays to all non-porous surfaces in the treatment zone.
- Dwell Time: Ensure surfaces remain visibly wet for the required duration to achieve total pathogen elimination.
Sterilization Standards for Reusable Tools
Any tool that penetrates the skin or contacts mucosal membranes must undergo a rigorous sterilization process.
Med spas must use steam sterilization equipment to ensure all microbial life, including fungal spores, is destroyed.
Cleaning instruments with soap and water is only the first step; it does not replace the need for heat-based sterilization.
Reusable instruments should be packaged in sterilization pouches with chemical indicators that change color when the process is complete.
Sterilization logs must be maintained to document every cycle, including the date, time, and temperature reached.
Biological monitoring, such as spore testing, should be performed weekly to verify that the equipment is functioning correctly.
Cross-Contamination Prevention Strategies
Preventing cross-contamination requires a strict separation of “clean” and “dirty” areas within the med spa facility.
Single-use items, such as cotton swabs, gauze, and wooden applicators, must be disposed of immediately after one use.
Products should be dispensed into secondary containers using clean spatulas to avoid contaminating the original bulk supply.
Sharps Management and Biohazard Disposal
- Sharps Containers: All needles and lancets must be placed in puncture-resistant, leak-proof containers immediately after use.
- Biohazard Bags: Items saturated with blood or bodily fluids require disposal in labeled red biohazard bags.
- Safe Handling: Never recap needles by hand to prevent accidental needlestick injuries among clinical staff.
Staff Training and Compliance Monitoring
Infection control systems are only effective when all staff members follow the same standardized safety protocols.
Annual training on bloodborne pathogens and OSHA standards ensures that every team member understands their safety responsibilities.
Regular clinical audits help supervisors identify gaps in sanitation and implement corrective actions before safety is compromised.
Documenting every safety process provides a clear history of compliance and protects the facility during regulatory inspections.
Clear signage in sterilization rooms and treatment areas serves as a constant reminder of the high hygiene standards required.
Maintaining a culture of safety ensures that the med spa remains a low-risk environment for both staff and clients.