Med Spa Infection Control and Clinical Safety Standards
Med spa infection control is a systematic framework of sanitation, disinfection, and sterilization protocols designed to eliminate the transmission of infectious agents within an aesthetic facility. These OSHA-aligned standards protect practitioners and clients by establishing rigorous barriers against cross-contamination during non-invasive and minimally invasive cosmetic treatments in a clinical environment.
Regulatory Compliance and OSHA Alignment
Every med spa must operate under a formal exposure control plan. This document outlines how the facility manages bloodborne pathogens and hazardous materials used during aesthetic procedures.
Compliance requires regular staff training on universal precautions. Practitioners must treat all human blood and bodily fluids as if they are known to be infectious for bloodborne pathogens.
Standardized record-keeping for sterilization cycles and safety data sheets ensures the facility meets state board and federal safety expectations. Documentation is the foundation of clinical accountability.
Multi-Level Disinfection and Sterilization Protocols
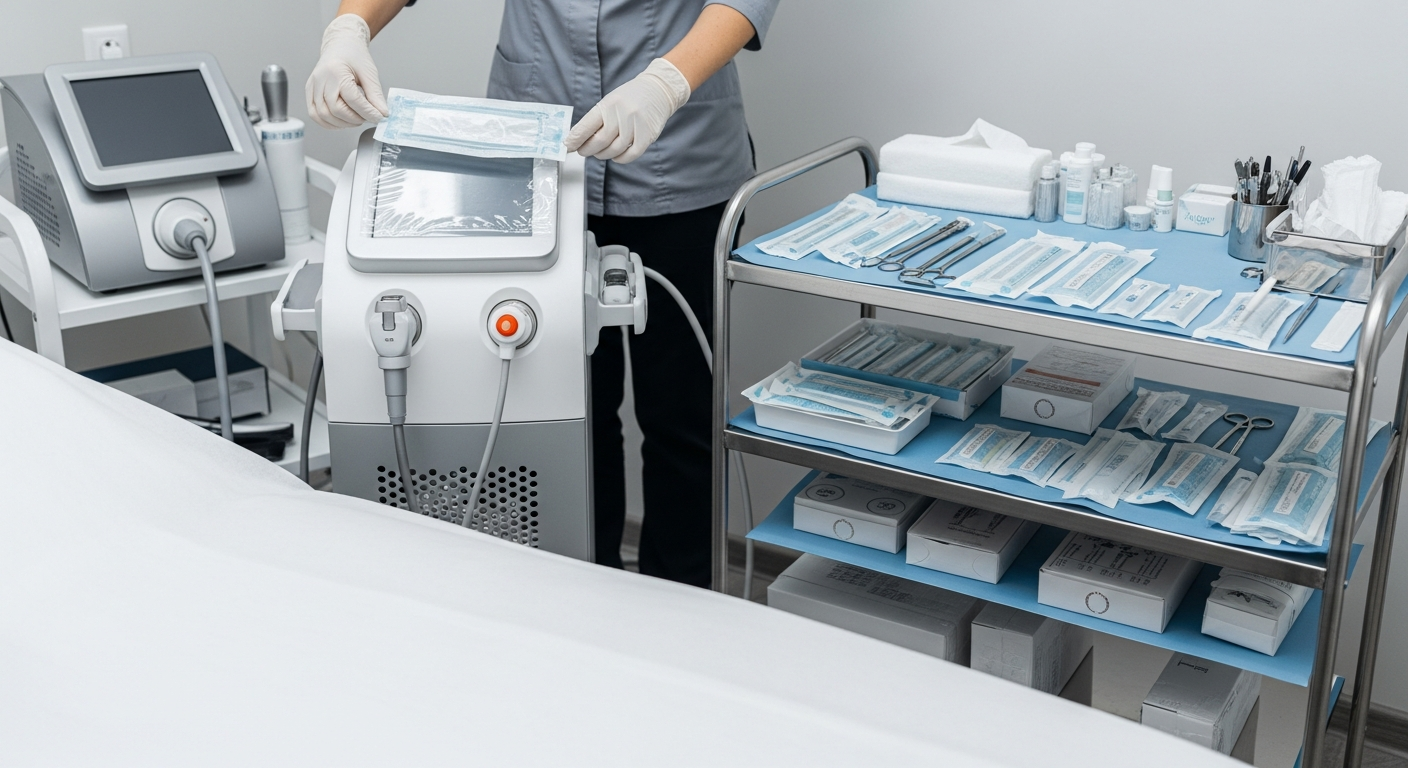
Treatment Room Surface Disinfection
Clinical surfaces must be cleaned and disinfected between every client. This includes treatment tables, counters, and high-touch equipment such as laser handpieces and device screens.
Practitioners must use EPA-registered, intermediate-level disinfectants. These solutions must remain wet on the surface for the full manufacturer-recommended contact time to ensure efficacy.
Disposable barriers are utilized for equipment that is difficult to decontaminate. These single-use covers are discarded immediately after the treatment is completed to maintain a sterile field.
Instrument Processing and Sterilization
Critical instruments that penetrate the skin or contact sterile tissue must undergo a complete sterilization process. This typically involves ultrasonic cleaning followed by pressurized steam sterilization.
Autoclaves must be monitored using chemical indicators and weekly biological spore tests. These tests provide clinical proof that the equipment has reached the required temperature and pressure.
Sterilized tools must remain in sealed, dated pouches until the moment of use. If a pouch is punctured or damp, the instruments are no longer considered sterile.
Cross-Contamination Prevention Strategies
The layout of a med spa should facilitate a one-way flow from contaminated to clean areas. This physical separation prevents soiled instruments from contacting sterile supplies.
Hand hygiene is the most effective way to prevent the spread of infection. Practitioners must wash hands before donning gloves and immediately after glove removal.
Single-use items, such as needles, lancets, and cotton swabs, must never be reused. Sharp items are disposed of in puncture-resistant containers located at the point of use.
Practitioner Hygiene and Personal Protective Equipment
Personal Protective Equipment (PPE) serves as a vital barrier between the clinician and potential pathogens. Required gear often includes clinical gloves, masks, and eye protection.
Gloves must be changed between every client and during a single procedure if they become torn or heavily soiled. PPE is never worn outside of the designated treatment area.
Professional attire should be dedicated to the clinical environment. Lab coats or scrubs provide an additional layer of protection and should be laundered using high-temperature cycles.
Post-Treatment Room Turnover
Room turnover is a timed process that ensures the environment is safe for the next client. All waste must be segregated into general trash or biohazardous containers according to local regulations.
Once the room is cleared of disposables, all non-porous surfaces undergo a thorough wipe-down. The room must remain vacant for the required disinfectant dwell time before the next entry.
Regular auditing of these turnover steps helps management maintain a consistent standard of safety across all providers. Systematic execution prevents human error in the sanitation chain.