Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic implementation of sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination in aesthetic clinics. These practices include OSHA-aligned bloodborne pathogen training, rigorous treatment room decontamination, and the use of EPA-registered disinfectants to maintain a sterile environment for non-surgical cosmetic procedures.
Core Sanitation Protocols for Treatment Rooms
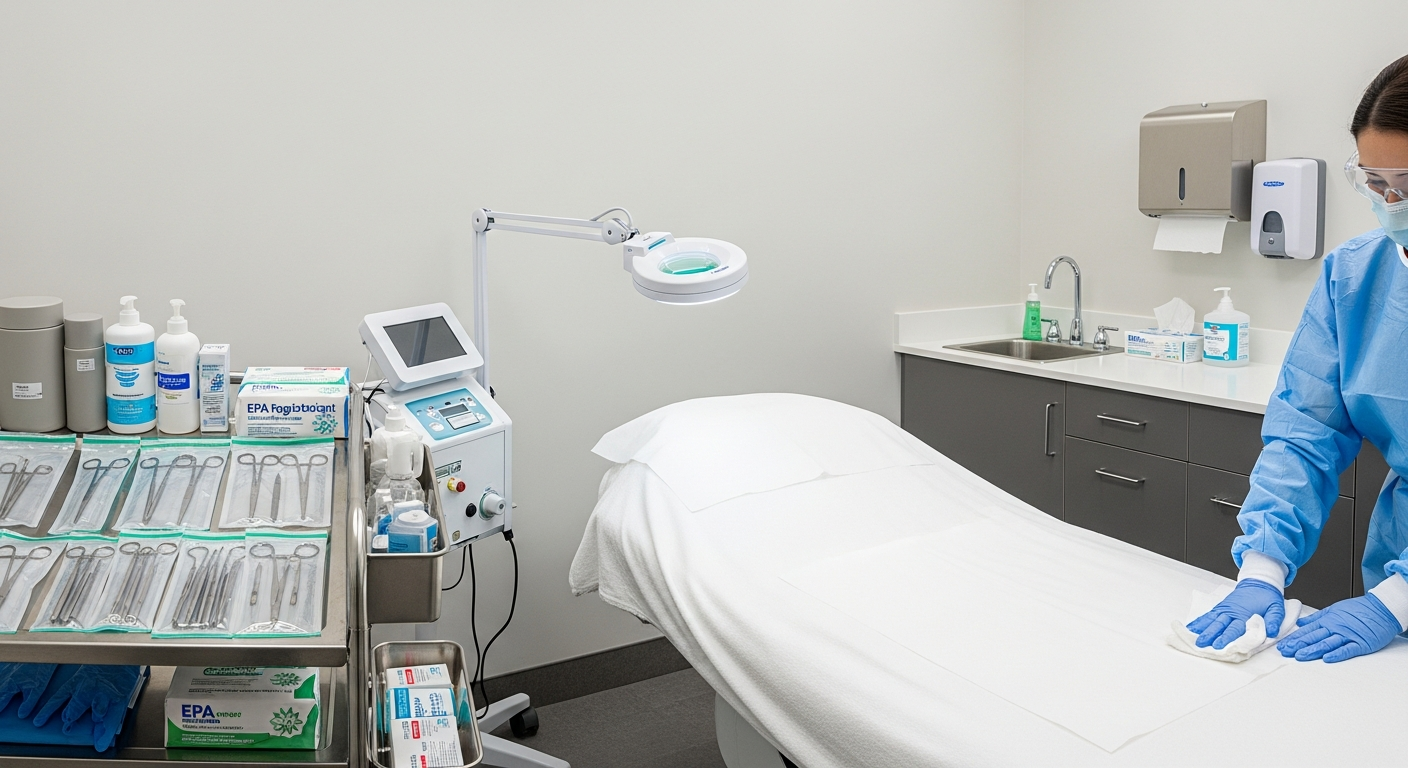
Maintaining a sterile environment begins with systematic treatment room management. Every surface must be treated as a potential source of contamination between client appointments.
Clinical staff must use EPA-registered, hospital-grade disinfectants on all high-touch surfaces. This includes treatment tables, counters, magnifying lamps, and product bottles used during the procedure.
All linens and disposable barriers must be replaced after every session. Single-use items should be discarded immediately in appropriate waste containers to prevent environmental exposure.
Sterilization Standards for Clinical Tools
Sterilization is a mandatory process for any reusable tool that penetrates the skin or contacts mucosal membranes. This process goes beyond basic cleaning or high-level disinfection.
Autoclave Maintenance and Testing
- Weekly Spore Testing: Facilities must perform biological indicator tests to verify the autoclave is reaching the required temperature and pressure.
- Proper Packaging: Tools must be dried and sealed in sterilization pouches with chemical indicators that change color upon successful processing.
- Storage Protocols: Sterilized items must remain in sealed pouches and be stored in clean, dry cabinets until the moment of use.
Management of Single-Use Devices
Many med spa treatments utilize disposable tips or needles. These items must never be cleaned or reused, regardless of the sterilization technology available on-site.
OSHA Compliance and Personal Protective Equipment
Occupational Safety and Health Administration (OSHA) standards protect both the provider and the client. Compliance requires a written exposure control plan and regular staff training.
Personal Protective Equipment (PPE) serves as the primary barrier against bloodborne pathogens. Clinicians must wear fresh gloves for every procedure and utilize eye protection during fluid-intensive treatments.
Proper sharps disposal is a critical component of med spa safety. All needles, syringes, and lancets must be placed in puncture-resistant, leak-proof containers labeled with biohazard symbols.
Cross-Contamination Prevention Systems
Cross-contamination occurs when pathogens are transferred from one surface or person to another. Rigid operational systems are necessary to break this chain of infection.
Hand hygiene remains the most effective way to prevent the spread of germs. Staff must wash hands with antimicrobial soap before donning gloves and immediately after glove removal.
Product dispensing protocols should prevent “double-dipping.” Use sterile spatulas or disposable applicators to remove creams or gels from bulk containers to maintain product integrity.
Documentation and Safety Audits
Consistency in infection control is verified through rigorous record-keeping. Med spas should maintain logs for equipment maintenance, sterilization cycles, and staff health safety training.
Internal safety audits help identify gaps in sanitation before they lead to clinical issues. Regular reviews of treatment room turnover times ensure staff are not rushing through disinfection steps.
By prioritizing these systematic processes, aesthetic clinics ensure a high standard of clinical safety that protects the health of both the practitioners and the public.