Med Spa Infection Control: Essential Clinical Standards
Med spa infection control is the systematic application of sanitation, disinfection, and sterilization protocols to prevent pathogen transmission within aesthetic clinical settings. These procedures align with OSHA standards to protect clients and practitioners during skin-rejuvenation and injectable treatments by maintaining rigorous hygiene across all equipment and treatment room surfaces.
OSHA-Aligned Sanitation Protocols
Every med spa must establish a written exposure control plan that follows federal safety guidelines. This document outlines how the facility manages bloodborne pathogens and chemical hazards during daily operations.
Standard precautions require staff to treat all body fluids as potentially infectious. This mindset ensures a high baseline of safety regardless of the specific treatment being performed.
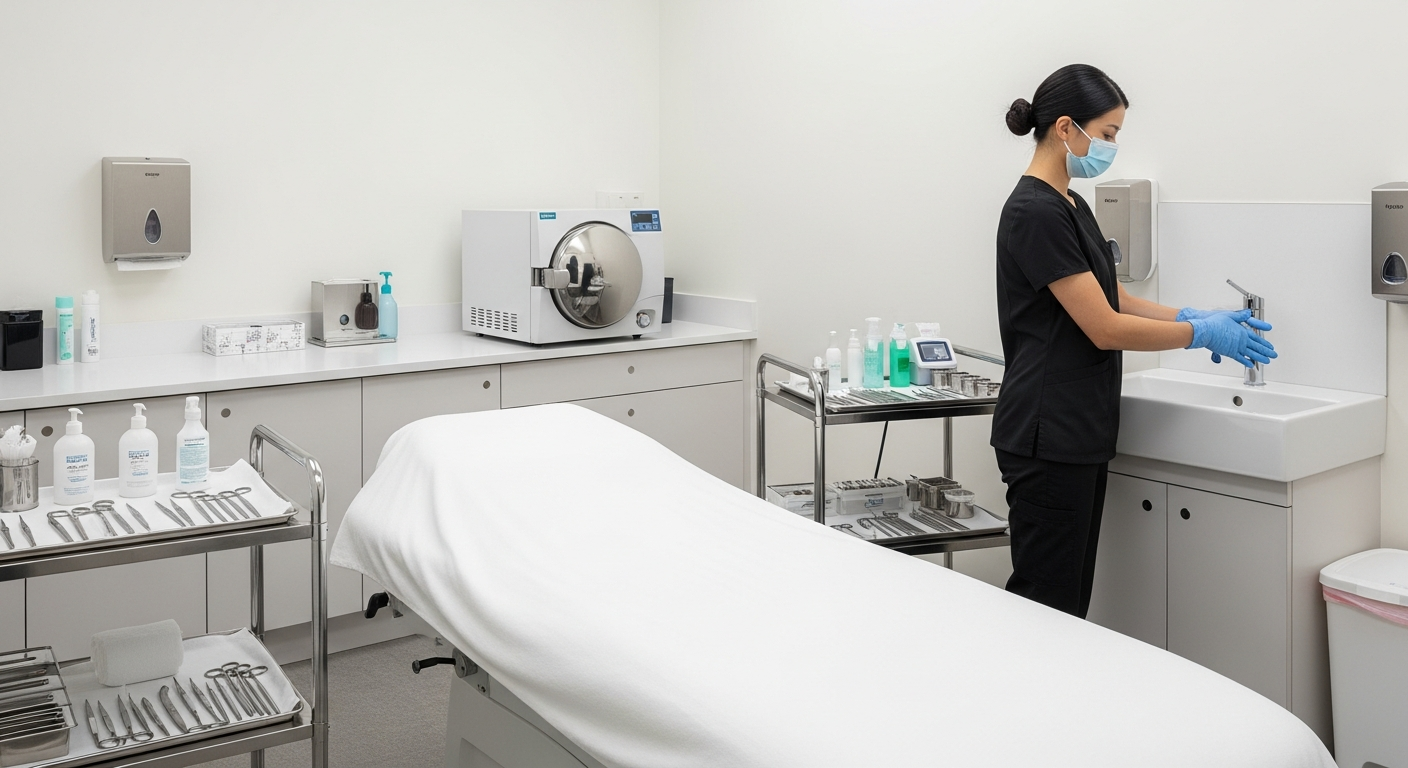
Hand hygiene remains the primary defense against infection. Clinical staff must wash hands before and after every client contact and whenever gloves are changed between procedures.
Treatment Room Disinfection Systems
Clinical surfaces must be cleaned and disinfected between every client session. This includes treatment tables, countertops, and any magnifying lamps or equipment touched during a procedure.
Practitioners must use EPA-registered disinfectants that are specifically rated for healthcare environments. These solutions must remain wet on surfaces for the full contact time recommended by the manufacturer.
Porosity dictates the level of cleaning required. Non-porous surfaces are easily wiped down, while porous items should be single-use or protected by impervious barriers to prevent contamination.
Surface Barrier Management
Using disposable barriers on high-touch areas reduces the reliance on chemical cleaners alone. Items like plastic sleeves for power cords and paper covers for tables provide an extra layer of protection.
Barriers must be discarded immediately after the treatment ends. The underlying surface should still be wiped down if the barrier was breached or moved during the session.
Sterilization Standards for Reusable Tools
Any instrument that penetrates the skin or contacts sterile tissue must undergo a full sterilization process. This typically involves a pressurized steam autoclave to ensure all microbial life is destroyed.
Reusable tools must be thoroughly cleaned of all organic debris before being placed in the sterilizer. Soil left on an instrument can act as a shield, preventing the steam from reaching pathogens.
Med spas must perform regular biological monitoring, such as weekly spore tests. These tests confirm that the sterilization equipment is functioning correctly and reaching the necessary temperature and pressure.
Cross-Contamination Prevention Strategies
Organizing the treatment room into “clean” and “dirty” zones prevents the accidental transfer of bacteria. Clean supplies should never be stored in areas where used instruments are processed.
Single-use items are the gold standard for preventing cross-contamination in a med spa. Needles, gauze, and applicators must be disposed of in appropriate sharps containers or biohazard bins immediately.
Practitioners must avoid reaching into multi-use product containers with gloved hands that have touched the client. Using sterile spatulas or portioning products into small cups prevents bulk contamination.
Personal Protective Equipment (PPE) Systems
Clinical supervisors must ensure that all staff have access to appropriate PPE, including medical-grade gloves, masks, and eye protection. PPE acts as a barrier between the provider and potential pathogens.
Gloves must be changed if they become torn or heavily soiled during a procedure. Proper removal techniques are essential to ensure the practitioner does not contaminate their skin during the process.