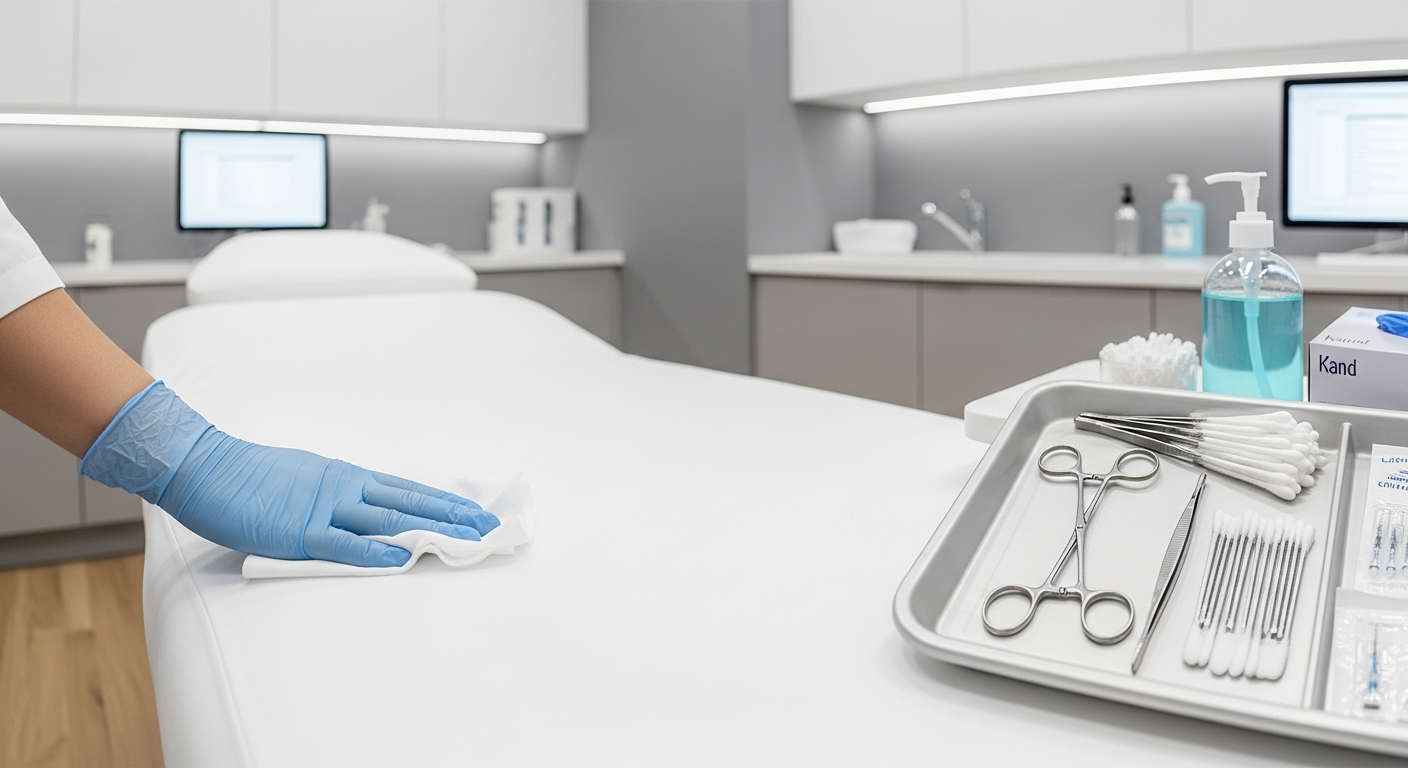
Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination and pathogen transmission within aesthetic clinics. These OSHA-aligned practices protect both clients and practitioners by ensuring treatment rooms, medical devices, and high-touch surfaces remain free from infectious agents during aesthetic procedures.
Core Components of Med Spa Sanitation
Standard precautions are the foundation of clinical safety in any aesthetic environment.
These protocols assume that all blood and body fluids are potentially infectious.
Med spas must implement consistent barriers to prevent the spread of pathogens.
Hand hygiene remains the single most effective way to prevent infection transmission.
Clinicians must wash hands thoroughly before and after every patient contact.
Alcohol-based hand rubs are acceptable when hands are not visibly soiled.
Personal Protective Equipment (PPE) provides a vital physical barrier during treatments.
Gloves, masks, and eye protection should be selected based on the specific procedure.
Single-use PPE must be disposed of immediately after each patient encounter.
Sterilization Standards for Aesthetic Tools
Any instrument that penetrates the skin or contacts mucous membranes requires sterilization.
Critical items must undergo a validated sterilization process, such as pressurized steam.
Documentation of every sterilization cycle is mandatory for compliance and safety monitoring.
Semi-critical items contact intact mucous membranes but do not penetrate the skin.
These tools require high-level disinfection at a minimum to ensure safety.
Most med spas prefer using disposable, single-use versions of these tools when possible.
Storage of sterilized instruments must prevent re-contamination before the next use.
Sterilized packs should be kept in clean, dry cabinets away from moisture.
Integrity of the packaging must be verified before any tool is used on a client.
Treatment Room Disinfection Systems
Treatment surfaces must be cleaned and disinfected between every client visit.
Environmental Protection Agency (EPA) registered disinfectants are required for medical surfaces.
The contact time specified on the disinfectant label must be strictly followed.
High-touch areas like equipment dials, stools, and countertops require frequent attention.
Using disposable barriers on difficult-to-clean surfaces can reduce the risk of contamination.
These barriers must be replaced between every patient to maintain a sterile field.
Floor and wall maintenance contributes to the overall reduction of the microbial load.
Clinical rooms should feature non-porous surfaces that withstand frequent chemical cleaning.
Fabric or carpeted surfaces are generally avoided in active treatment zones.
Preventing Cross-Contamination in Clinical Areas
Establishing clear “clean” and “dirty” zones prevents the mixing of supplies.
Contaminated instruments must be transported to the processing area in puncture-resistant containers.
Clean supplies should never be stored in the same area as used instruments.
Sharps safety is a critical component of preventing bloodborne pathogen exposure.
Needles and blades must be discarded in rigid, leak-proof containers immediately.
Sharps containers must be replaced when they reach the designated fill line.
Waste management protocols distinguish between general trash and regulated biohazardous waste.
Saturated gauze or items dripping with blood must be placed in biohazard bags.
Proper labeling and disposal through certified waste haulers ensure environmental safety.
Compliance and Safety Oversight
Regular staff training ensures that infection control protocols are followed consistently.
A designated safety officer should perform periodic audits of sanitation logs.
Updated Safety Data Sheets (SDS) must be accessible for all chemicals used on-site.
Systematic reviews of infection control manuals help med spas adapt to new guidelines.
Consistent documentation of training and sterilization cycles creates a culture of safety.
A proactive approach reduces clinical risks and ensures a professional treatment environment.