Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic implementation of sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination within aesthetic clinical environments. These OSHA-aligned practices ensure the safety of both practitioners and clients by establishing rigorous barriers against bloodborne pathogens and infectious agents during non-invasive and minimally invasive cosmetic procedures.
OSHA-Aligned Sanitation Practices for Providers
Effective infection control begins with rigorous hand hygiene and the appropriate use of personal protective equipment.
Practitioners must wash hands with antimicrobial soap before and after every client contact to prevent the transfer of microorganisms.
Gloves must be changed between every procedural step involving different anatomical sites or contaminated surfaces.
Personal protective equipment acts as a critical physical barrier in the med spa environment.
Face masks and eye protection should be utilized during any treatment where splashing or aerosolization of fluids is possible.
Proper disposal of single-use items in designated biohazard containers is mandatory to maintain a sterile workspace.
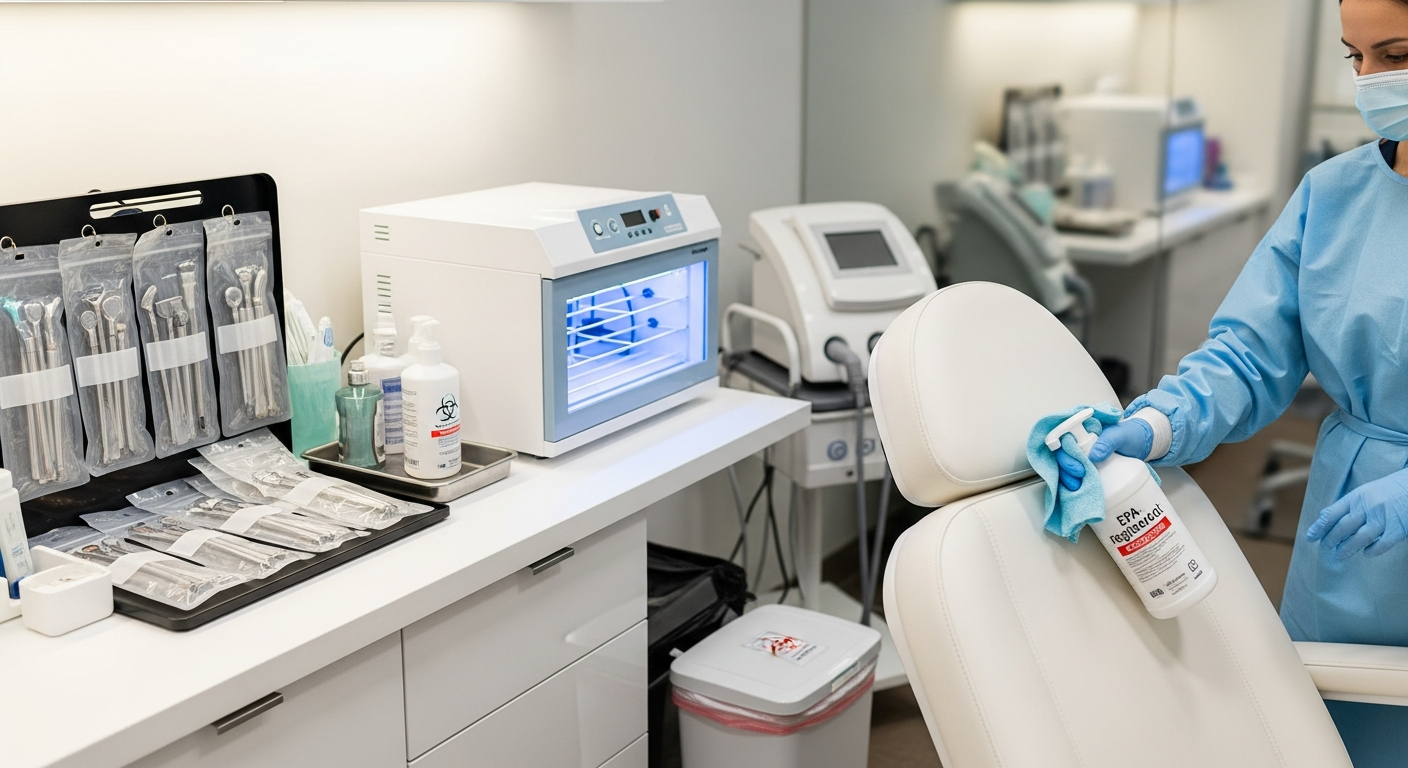
Treatment Room Disinfection Systems
Every treatment room requires a systematic decontamination process between every appointment.
Environmental surfaces, including treatment tables and equipment leads, must be cleaned with EPA-registered disinfectants.
These solutions must remain wet on the surface for the full manufacturer-recommended contact time to ensure efficacy.
High-touch areas such as door handles, counters, and magnifying lamps require frequent periodic cleaning throughout the day.
Using disposable barriers on equipment interfaces can further reduce the risk of cross-contamination.
A documented cleaning log helps supervisors track compliance and ensure no steps are bypassed during busy shifts.
Sterilization Standards for Reusable Implements
Any instrument that penetrates the skin or contacts mucosal membranes must undergo high-level sterilization.
This process typically involves thorough ultrasonic cleaning to remove debris followed by pressurized steam sterilization.
Biological indicators must be used regularly to verify that the sterilization equipment is functioning at the correct parameters.
Storage of sterilized tools is just as critical as the sterilization process itself.
Instruments must remain in sealed, dated pouches until the exact moment of use during a treatment.
If a pouch is punctured or damp, the items inside are no longer considered sterile and must be reprocessed.
Cross-Contamination Prevention Strategies
Cross-contamination is prevented by strictly separating clean and soiled utility areas within the med spa.
Dirty implements should never be transported through common areas without being placed in a leak-proof container.
Supplies should be dispensed using the “no-touch” method or sterile applicators to avoid contaminating bulk product containers.
Standard precautions assume that all body fluids are potentially infectious regardless of a client’s health history.
Sharps containers must be placed within arm’s reach of the treatment area to facilitate safe disposal of needles.
Consistent adherence to these workflows creates a culture of safety that protects both the staff and the public.