Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic protocols and sanitation practices used to prevent the spread of pathogens and ensure client safety during aesthetic treatments. These standards include proper hand hygiene, treatment room disinfection, and the sterilization of reusable instruments to maintain a sterile clinical environment and comply with regulatory requirements.
Regulatory Alignment and OSHA Standards
Every facility must align its internal operations with federal and state safety guidelines.
OSHA standards require med spas to maintain a clean environment to protect both staff and clients.
A written exposure control plan is necessary to manage biological risks effectively.
Bloodborne Pathogen Compliance
Med spa staff must receive annual training on handling potential bloodborne pathogens.
This includes the correct disposal of sharps and contaminated materials in labeled containers.
Proper management of these materials prevents accidental needle-stick injuries and cross-contamination.
Treatment Room Disinfection Protocols
Standardized cleaning cycles must occur between every client appointment.
All high-touch surfaces, including treatment tables and equipment handles, require thorough sanitation.
Consistency in these routines ensures that no pathogens remain after a procedure is completed.
Surface Sanitation and Contact Times
Clinical-grade disinfectants must be used according to the manufacturer’s specified contact time.
The surface must remain wet for the full duration to ensure the elimination of bacteria and viruses.
Wiping away a solution too early can result in incomplete disinfection of the treatment area.
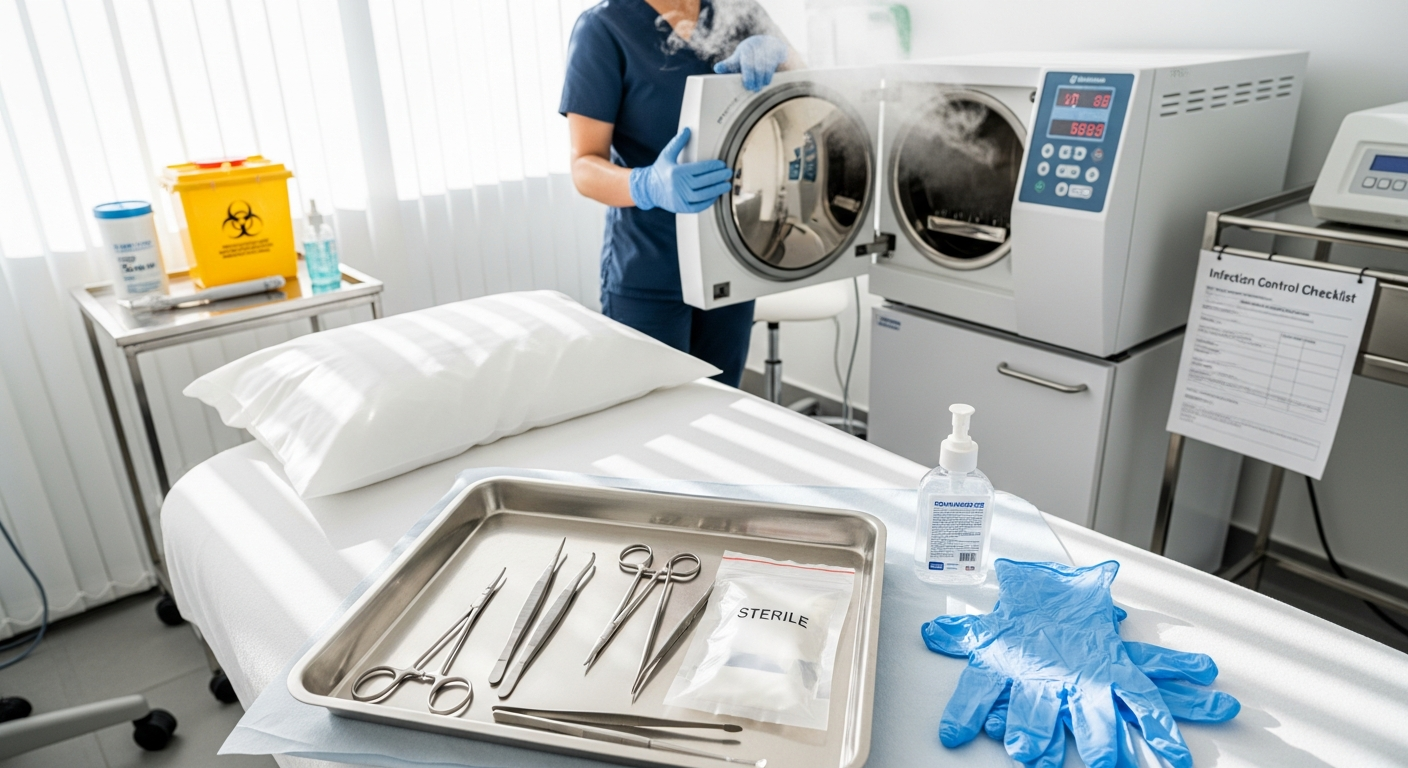
Sterilization and Instrument Processing
Reusable tools must undergo a multi-step cleaning and sterilization process.
Initial cleaning removes organic debris before instruments enter a high-level sterilization unit.
This process is critical for any tool that penetrates or contacts broken skin during treatments.
Autoclave Management and Testing
Sterilization equipment requires regular maintenance and biological monitoring to ensure efficacy.
Spore testing should be performed weekly to confirm the autoclave is reaching required temperatures.
Detailed logs of these tests serve as proof of compliance and clinical safety.
Cross-Contamination Prevention Systems
Preventing the transfer of pathogens requires strict adherence to barrier techniques.
Practitioners must treat every treatment room as a controlled clinical environment.
Separating clean and soiled supplies is a fundamental requirement for operational safety.
Personal Protective Equipment Usage
Personal protective equipment, or PPE, serves as a physical barrier against fluids and debris.
Gloves must be changed between every client and after touching non-sterile surfaces.
Masks and eye protection provide additional safety during procedures that may generate splashes.
Hand Hygiene and Practitioner Safety
Hand hygiene remains the most effective way to prevent the spread of infections.
Staff must wash hands before donning gloves and immediately after removing them.
This practice protects the practitioner and maintains the integrity of the clinical field.
-
Core Safety Requirements:
- Use of EPA-registered disinfectants for all surface cleaning.
- Consistent use of single-use disposables whenever possible.
- Mandatory sharps container placement in every treatment room.
- Strict adherence to instrument sterilization logs and testing.