Med Spa Infection Control: Clinical Sanitation Standards
Med spa infection control is a systematic approach to preventing pathogen transmission through rigorous sanitation, sterilization, and disinfection protocols within aesthetic clinical environments. These OSHA-aligned practices protect clients and staff by establishing standardized workflows for treatment room preparation, tool processing, and the management of bloodborne pathogens during non-invasive cosmetic procedures.
Standardized Sanitation and Disinfection Protocols
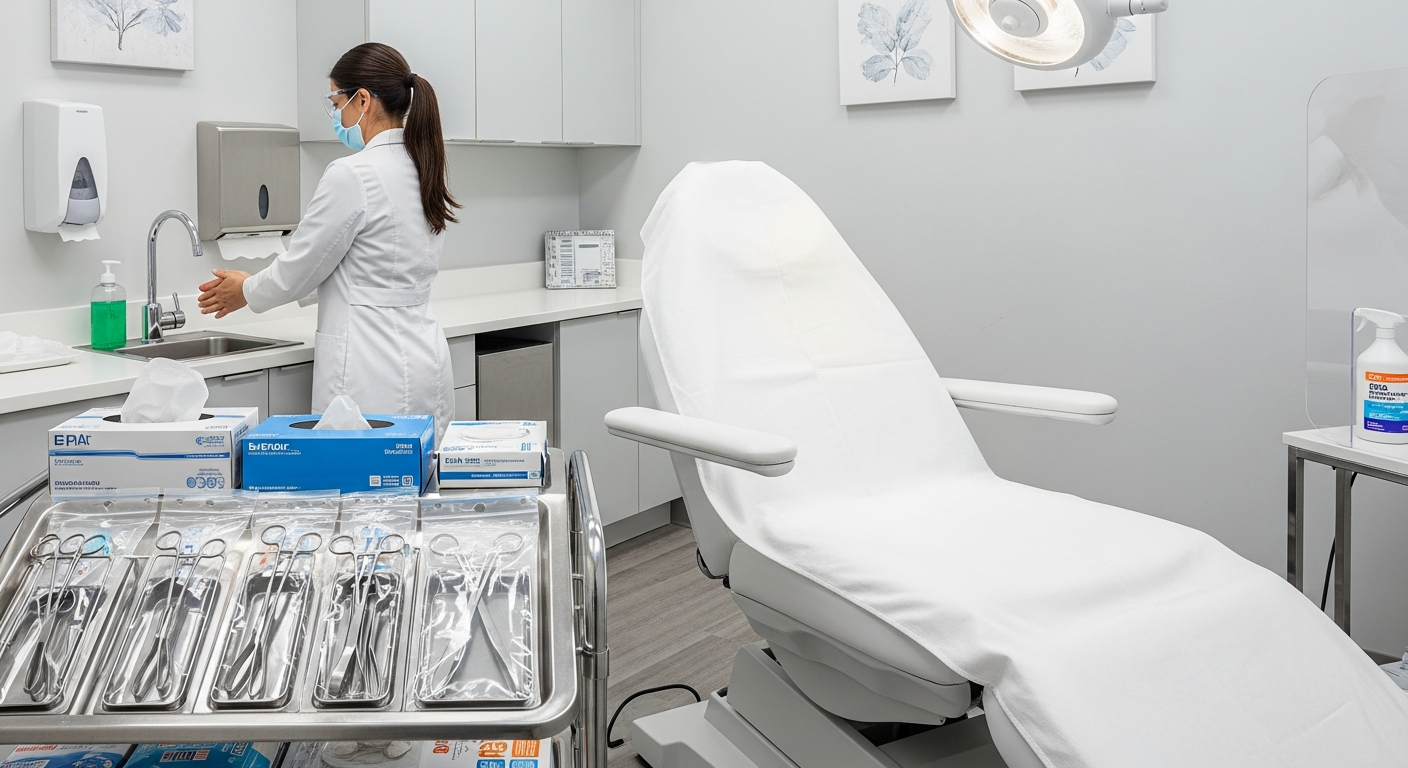
Every med spa must implement a multi-tiered sanitation system to maintain a sterile environment. This begins with the use of EPA-registered, hospital-grade disinfectants on all hard surfaces.
Treatment tables, work carts, and practitioner stools must be disinfected between every client. Staff should allow for the specific contact time listed on the disinfectant label to ensure efficacy.
Treatment Room Workflow Management
Establishing a “clean to dirty” workflow prevents the spread of contaminants throughout the facility. All supplies needed for a procedure should be laid out on a sanitized tray before the client enters.
High-touch surfaces, including light switches and magnifying lamps, should be covered with disposable plastic barriers. These barriers must be replaced after every treatment session to eliminate cross-contamination.
Sterilization Standards for Reusable Instruments
Any reusable tool that penetrates the skin or comes into contact with bodily fluids requires high-level sterilization. This process moves beyond simple cleaning to the total destruction of microbial life.
Mechanical cleaning with water and detergent must occur before sterilization to remove organic debris. This ensures the sterilization agent can effectively reach all surfaces of the instrument.
Autoclave Operation and Monitoring
Steam sterilization via an autoclave is the industry standard for med spa instrument processing. Devices must be operated according to the manufacturer’s pressure and temperature requirements.
Clinical supervisors should perform weekly biological indicator tests to verify the autoclave is functioning correctly. Internal chemical indicators must also be used in every sterilized pack to confirm exposure to heat.
OSHA Alignment and Bloodborne Pathogen Safety
Med spa operations in the United States must comply with OSHA Bloodborne Pathogens Standards. This includes the mandatory use of Personal Protective Equipment (PPE) such as gloves, masks, and eye protection.
A written Exposure Control Plan is required to outline how the facility manages needle sticks or fluid splashes. This document must be accessible to all staff members at all times.
Sharps Management and Disposal
Proper disposal of needles and lancets is a critical component of med spa infection control. All sharps must be placed in puncture-resistant, leak-proof containers immediately after use.
Sharps containers should be replaced when they reach the designated fill line to prevent accidental injury. Waste must be handled by a licensed biohazard disposal service in accordance with local regulations.
Cross-Contamination Prevention Strategies
Hand hygiene is the single most effective way to prevent the spread of infections in a med spa. Practitioners must wash hands with soap and water before donning and after doffing gloves.
Single-use items, such as cotton swabs and wooden spatulas, must never be “double-dipped” into multi-use product containers. Use a sterile secondary container for products to maintain the integrity of the original supply.
Staff Training and Safety Documentation
Regular training ensures that all clinical staff understand the latest infection control techniques. Training sessions should cover hand hygiene, PPE usage, and the proper dilution of cleaning chemicals.
Detailed logs must be maintained for all sanitation activities, including equipment maintenance and sterilization cycles. These records provide a verifiable audit trail for compliance managers and regulatory inspectors.