Essential Med Spa Regulations and Compliance Requirements
Med spa regulations are state-specific legal frameworks governing the operation of aesthetic medical clinics, focusing on professional licensing, corporate practice of medicine, and medical supervision. These laws ensure that clinical procedures, like injectables and laser treatments, are performed by qualified practitioners under the direct or indirect oversight of a licensed medical director.
State-Specific Legal Frameworks and CPOM
In the United States, med spa regulations vary significantly by state. Most jurisdictions adhere to the Corporate Practice of Medicine (CPOM) doctrine.
This doctrine generally prohibits non-physicians from owning a medical practice or employing physicians to provide clinical services.
To remain compliant, many owners utilize a Management Services Organization (MSO) model to separate business operations from clinical care.
Medical Supervision and Licensing Requirements
Every med spa must operate under the license of a Medical Director, typically a board-certified physician or, in some states, an NP.
The Medical Director is responsible for clinical protocols, patient safety, and ensuring that all staff act within their legal scope.
Supervision levels—classified as direct, indirect, or general—dictate whether the physician must be physically present during medical procedures.
Aesthetic Scope of Practice and Delegation
Compliance requires a strict understanding of which professionals can legally perform specific aesthetic treatments.
While Registered Nurses (RNs) often perform injectables, they must do so under a physician’s order following a Good Faith Exam.
Estheticians are generally limited to non-invasive skin care and cannot legally perform “medical acts” like deep chemical peels or injections.
The Importance of the Good Faith Exam
A Good Faith Exam (GFE) is a mandatory clinical evaluation performed before a patient receives their first medical aesthetic treatment.
The exam must be conducted by a physician, Nurse Practitioner, or Physician Assistant to establish a formal provider-patient relationship.
Failure to perform and document a GFE is a frequent cause of regulatory disciplinary action and professional license investigations.
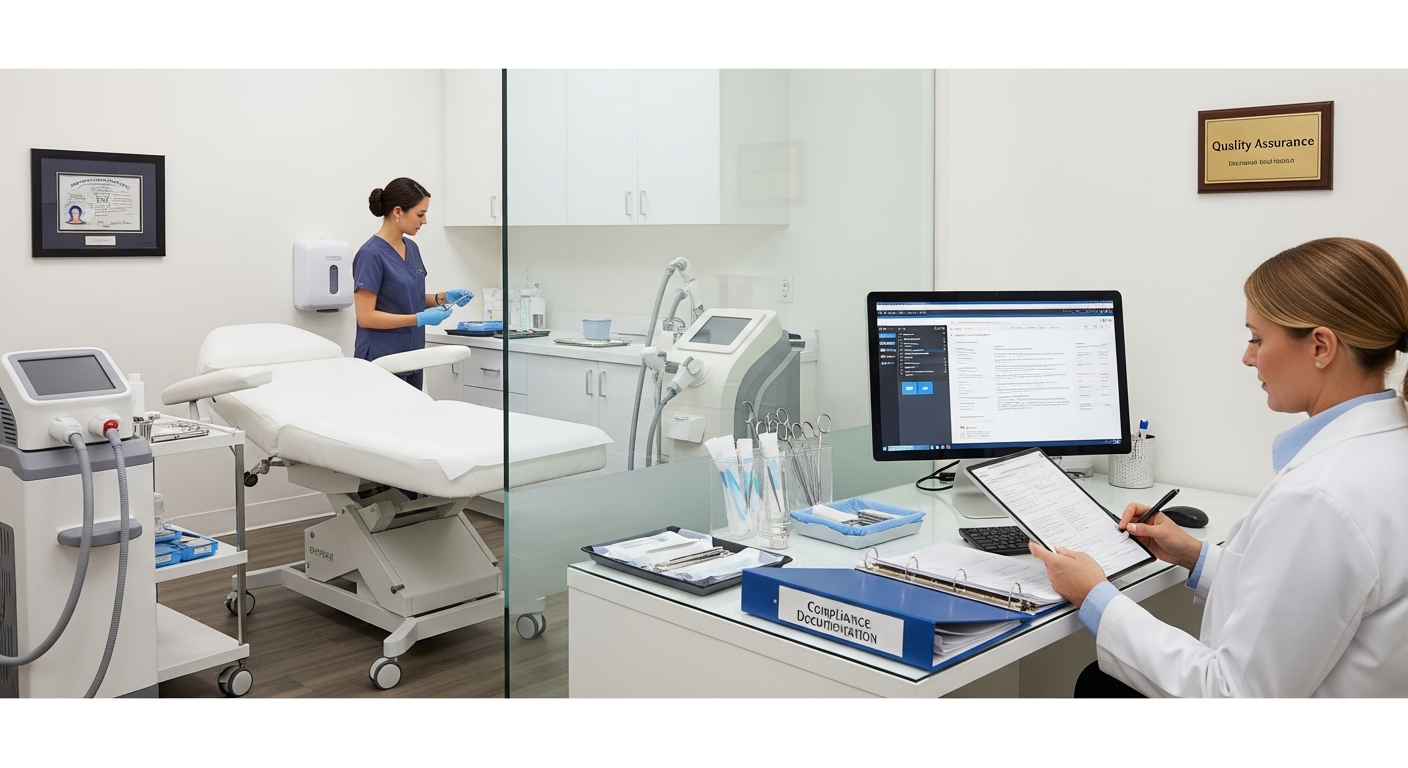
Inspection Readiness and Compliance Documentation
State boards of medicine and nursing may conduct unannounced inspections to verify that a facility meets healthcare safety standards.
Essential compliance documentation includes:
- Written Standard Operating Procedures (SOPs) for every medical treatment.
- Current professional licenses for all clinical and administrative staff.
- Evidence of proper hazardous waste disposal and OSHA compliance.
- Detailed patient charts that include informed consent and treatment logs.
HIPAA and Patient Privacy Standards
Because med spas provide medical services, they are “covered entities” under the Health Insurance Portability and Accountability Act (HIPAA).
Facilities must protect electronic protected health information (ePHI) through encrypted communication and secure digital record-keeping systems.
Staff training on patient privacy is a mandatory component of a comprehensive med spa compliance program.