Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent the spread of pathogens during aesthetic treatments. These OSHA-aligned practices ensure facility safety by establishing strict guidelines for hand hygiene, treatment room preparation, and the management of single-use and reusable clinical tools.
Core Principles of Med Spa Sanitation
Standard precautions form the basis of a safe clinical environment.
These protocols assume all blood and body fluids are potentially infectious.
Clinicians must maintain rigorous hand hygiene before and after every treatment.
Establishing a culture of safety requires clear, written operating procedures.
Staff must be trained on the specific chemistry of cleaning agents.
Regular audits help ensure every team member follows these safety steps.
Treatment Room Disinfection Protocols
Every treatment room requires a systematic cleaning process between appointments.
All non-porous surfaces must be treated with EPA-registered disinfectants.
This includes treatment tables, counters, and any equipment touched during procedures.
High-touch surfaces like light switches and door handles need frequent attention.
Disinfectants must remain wet on the surface for the full required contact time.
Wiping a surface dry too early prevents the solution from killing pathogens.
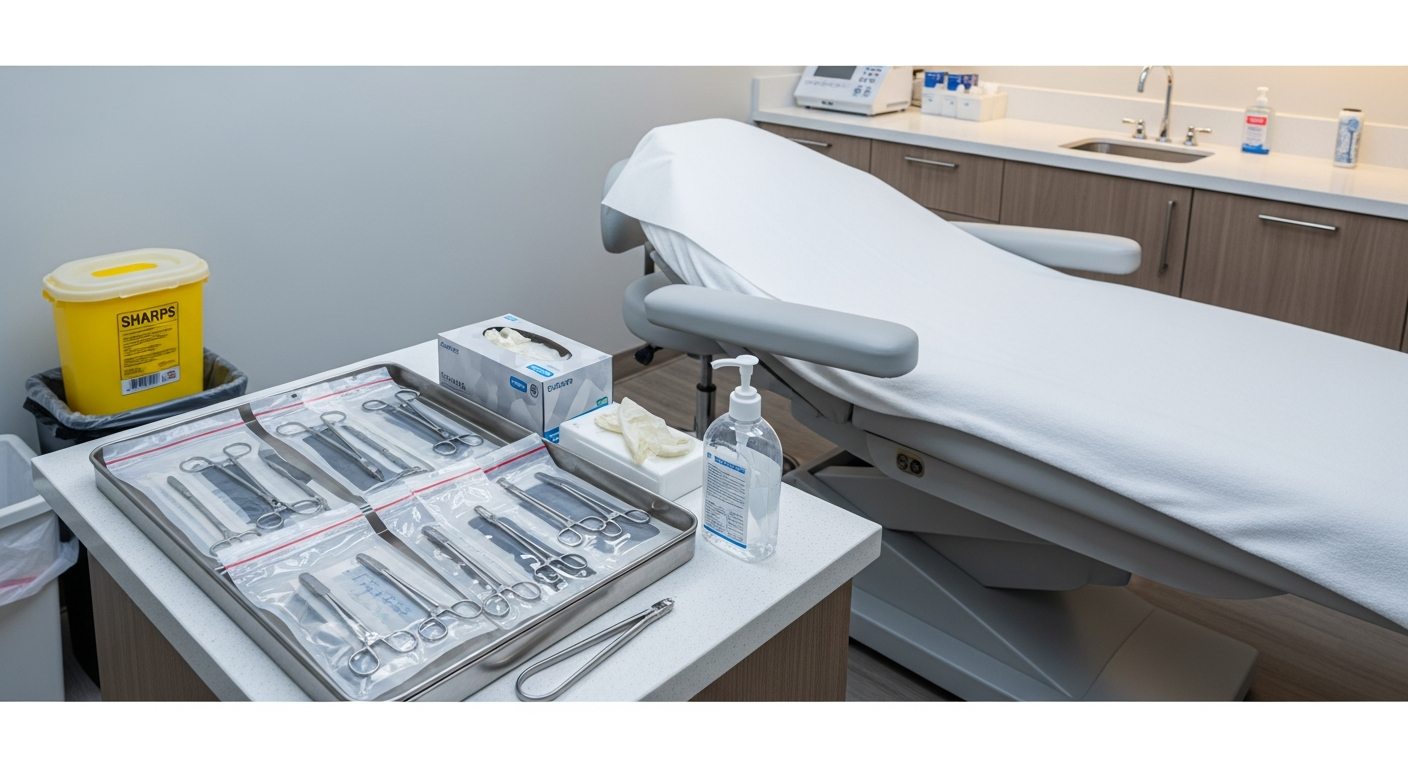
Sterilization Standards for Reusable Tools
Reusable instruments must undergo a multi-step decontamination process.
Tools are first cleaned to remove debris, then processed in a calibrated autoclave.
Proper biological monitoring ensures the sterilization equipment functions effectively every time.
Sterilized items must be stored in sealed pouches until the point of use.
If a pouch is torn or wet, the item is no longer considered sterile.
Always check chemical indicators to verify that the sterilization cycle was successful.
Safe Management of Clinical Sharps
Needles and lancets must be disposed of immediately in puncture-resistant containers.
Sharps containers should be replaced when they reach the designated fill line.
Never recap needles to prevent accidental needle-stick injuries among clinical staff.
Preventing Cross-Contamination in the Facility
Cross-contamination occurs when pathogens travel between surfaces, staff, and clients.
Effective prevention relies on the correct use of Personal Protective Equipment (PPE).
Gloves, masks, and eye protection create a barrier against potential infectious agents.
Clinicians should never touch clean supplies while wearing contaminated gloves.
Single-use items must be discarded immediately after a single client contact.
Consistency in these habits protects both the provider and the client.
Establishing Controlled Safety Zones
Designate specific areas for clean supplies and contaminated materials.
Ensure that staff traffic flow minimizes contact between dirty tools and sterile kits.
Regular audits of these zones help maintain high clinical safety standards.
-
Daily Safety Checklist Items:
- Verify autoclave cycle logs and biological test results.
- Ensure all treatment rooms are stocked with fresh PPE.
- Check expiration dates on all clinical solutions and supplies.
- Confirm sharps containers are not overfilled and are securely placed.