Med Spa Infection Control: Essential Clinical Protocols
Med spa infection control is the systematic application of sanitation, disinfection, and sterilization practices designed to eliminate pathogen transmission during aesthetic procedures. These protocols incorporate OSHA standards, rigorous treatment room decontamination, and the consistent use of personal protective equipment to maintain a sterile environment for both practitioners and clients within the facility.
Standard Precautions for Aesthetic Environments
Every med spa must operate under the principle of standard precautions. This approach treats all blood and bodily fluids as potentially infectious to minimize risk.
Hand Hygiene and Personal Protective Equipment
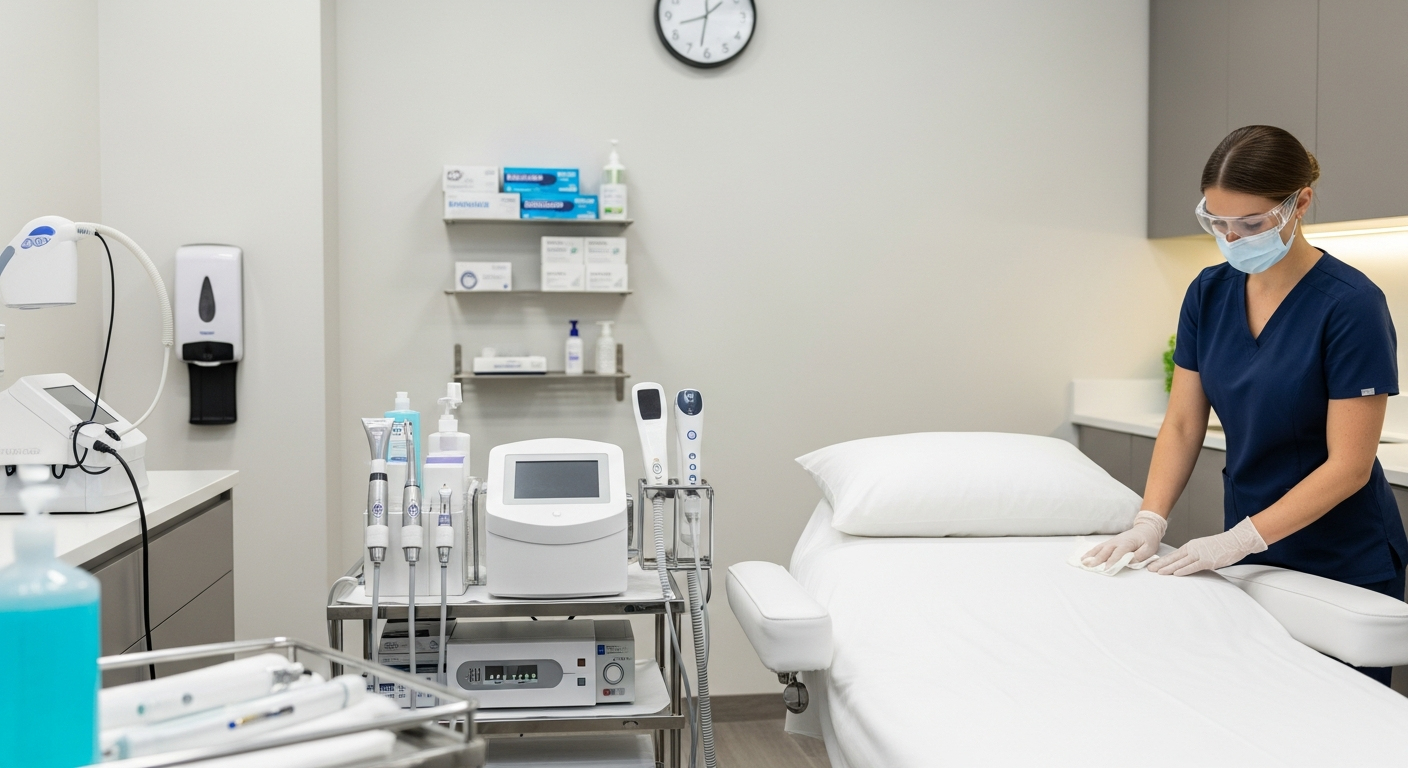
Clinicians must perform thorough handwashing before and after every client contact. Use liquid soap and water or high-grade sanitizers to prevent microbial transfer.
Personal protective equipment (PPE) acts as a critical barrier between the provider and pathogens. This includes disposable gloves, face masks, and protective eyewear during treatments.
Gloves must be changed between every client and immediately if they become punctured. Never reuse disposable PPE or attempt to sanitize single-use items.
Treatment Room Disinfection and Sanitation
The treatment room is the primary site for potential cross-contamination. Systematic cleaning schedules ensure that every surface remains clinical-grade and safe for use.
Surface Decontamination Procedures
High-touch surfaces such as treatment tables, counters, and stools require disinfection after every session. Use EPA-registered disinfectants approved for clinical settings.
Apply the “wipe-discard-wipe” method to ensure surfaces are physically cleaned and then chemically disinfected. Allow surfaces to remain wet for the full contact time.
Remove all porous items from the immediate treatment area. Items like linens and towels must be laundered at high temperatures with disinfecting agents after each use.
Sterilization and Device Management
Reusable tools require a higher level of processing than standard surfaces. Sterilization is mandatory for any implement that breaches the skin barrier or contacts mucous membranes.
Multi-Step Reprocessing Standards
The reprocessing cycle begins with pre-cleaning to remove organic debris. This step is vital because leftover material can shield pathogens from the sterilization process.
Mechanical cleaning should be followed by sterilization in a monitored autoclave. Maintain detailed logs of every cycle, including temperature, pressure, and biological indicator results.
Store sterilized instruments in sealed, dated pouches. If a pouch becomes wet or torn, the items inside are no longer considered sterile and must be reprocessed.
Preventing Cross-Contamination in Clinical Workflows
Cross-contamination often occurs through improper movement or handling of supplies. Establishing a clear flow between “clean” and “dirty” zones prevents the spread of bacteria.
Waste Management and Sharps Disposal
Biohazardous waste requires specialized handling and containment. Use leak-proof, color-coded bags for any materials saturated with blood or infectious fluids.
Sharps, including needles and lancets, must go into puncture-resistant containers immediately after use. Never recap needles, as this is a primary cause of accidental injury.
Regularly audit waste disposal areas to ensure compliance with local and federal regulations. Proper disposal protects staff, clients, and the surrounding community from exposure.