Med Spa Infection Control and Clinical Safety Standards
Med spa infection control refers to the systematic protocols and sanitation practices used to prevent the spread of pathogens within an aesthetic clinic. These procedures include high-level disinfection of treatment surfaces, proper sterilization of reusable instruments, and adherence to OSHA bloodborne pathogen standards to ensure a sterile environment for both practitioners and clients.
OSHA Compliance in Aesthetic Settings
Med spa infection control begins with strict adherence to Occupational Safety and Health Administration (OSHA) standards regarding bloodborne pathogens. Facilities must maintain a written exposure control plan that details specific safety measures and staff training requirements.
Personal protective equipment (PPE), including gloves, masks, and eye protection, serves as the primary barrier against accidental exposure. All clinical staff must receive annual training on handling sharps and hazardous materials safely to maintain a compliant environment.
Treatment Room Disinfection Protocols
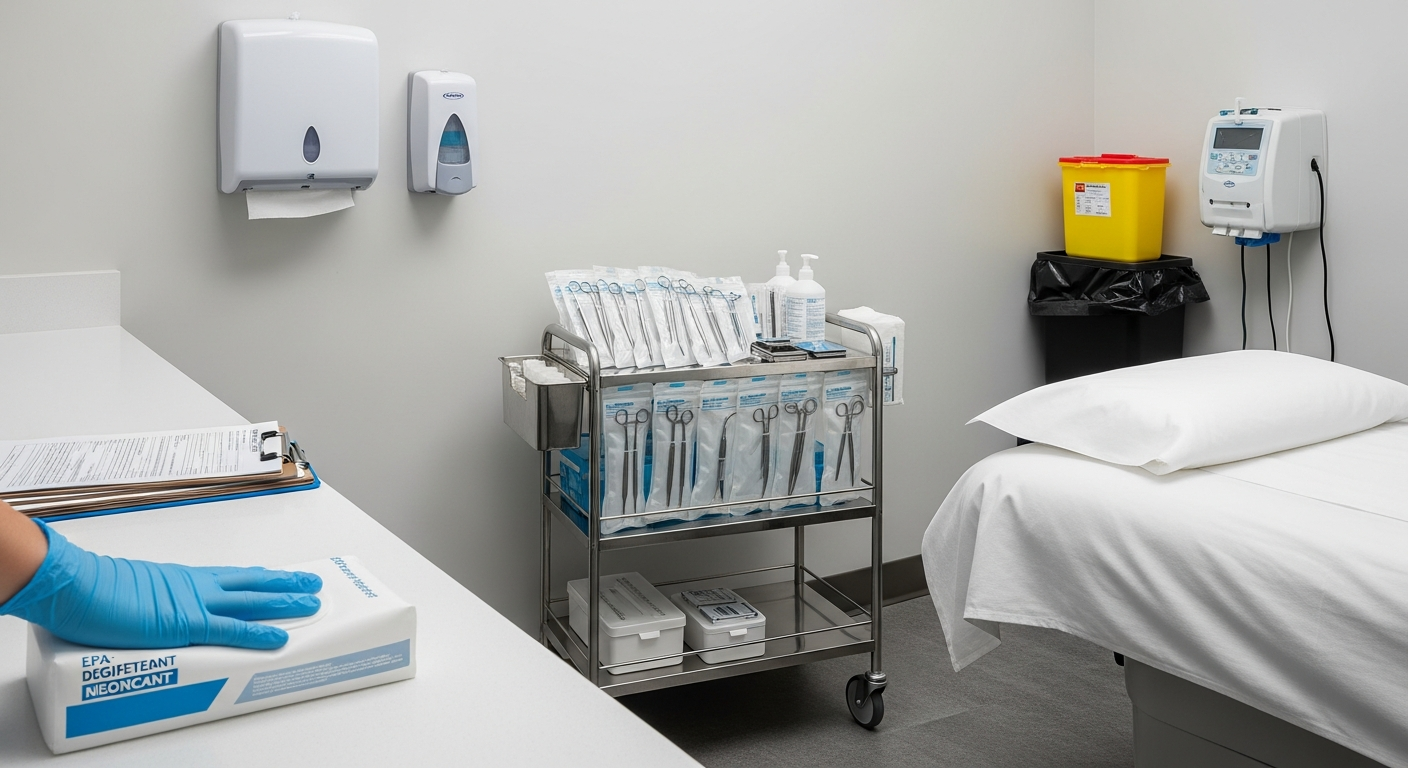
Systematic disinfection of treatment rooms must occur before and after every client encounter to eliminate cross-contamination risks. This involves using EPA-registered, medical-grade disinfectants on all non-porous contact surfaces within the treatment station area.
High-touch zones, such as treatment tables, stools, and product counters, require thorough saturation according to the disinfectant manufacturer’s contact time. Porous items or single-use supplies must be discarded immediately after use in appropriate clinical waste containers.
Sterilization Standards for Reusable Instruments
Any instrument that penetrates the skin or contacts mucosal membranes requires high-level sterilization, typically achieved via a monitored autoclave. Simple sanitation is insufficient for tools used in invasive or minimally invasive aesthetic procedures within a med spa.
Sterilization logs must track every cycle, including temperature, pressure, and biological indicator results to verify equipment efficacy. Instruments must remain in sealed, dated sterilization pouches until the exact moment of use during a scheduled clinical treatment.
Cross-Contamination Prevention Systems
Hand Hygiene and Sanitization
Rigorous hand hygiene is the most effective way to prevent the transmission of infections between clinical staff and clients. Practitioners must wash hands with antimicrobial soap or use high-alcohol sanitizer before donning and after doffing medical gloves.
Safe Supply Management
Storing supplies in closed cabinets prevents airborne contaminants from settling on clean tools or consumables during procedures. Multi-use containers must be accessed using sterile applicators to avoid contaminating the bulk product during active treatment sessions.
- Daily Logs: Maintain records of room disinfection and equipment sterilization cycles.
- Sharps Disposal: Utilize puncture-proof containers for all needles and blades.
- Biohazard Waste: Separate clinical waste from standard office refuse for proper disposal.
- Surface Barriers: Use disposable covers on equipment that cannot be easily disinfected.