Med Spa Infection Control: Clinical Safety Standards
Med spa infection control refers to the systematic application of sanitation, disinfection, and sterilization protocols designed to prevent pathogen transmission within aesthetic clinics. These practices include OSHA-aligned bloodborne pathogen training, treatment room surface disinfection, and instrument sterilization to ensure a safe environment for both practitioners and clients during non-invasive cosmetic procedures.
OSHA-Aligned Sanitation Protocols
Every med spa must establish a written exposure control plan that aligns with federal safety regulations.
This plan outlines how the facility manages bloodborne pathogens and occupational hazards.
Staff must undergo annual training to maintain compliance and clinical safety.
Personal Protective Equipment (PPE) Usage
Practitioners must use appropriate barriers such as medical-grade gloves, masks, and eye protection.
PPE must be changed between every client to prevent the transfer of microorganisms.
Hand hygiene remains the foundation of any effective infection control system.
Treatment Room Disinfection Systems
The treatment room environment requires high-level disinfection between every scheduled appointment.
All non-porous surfaces, including treatment tables and equipment carts, must be wiped down.
Using EPA-registered disinfectants ensures the elimination of bacteria, viruses, and fungi.
Surface Decontamination Procedures
Clinical staff should follow a “spray-wipe-spray” or “wipe-discard-wipe” technique for contact surfaces.
This ensures that organic debris is removed before the final disinfecting layer is applied.
Dwell times must be strictly followed according to the disinfectant manufacturer’s instructions.
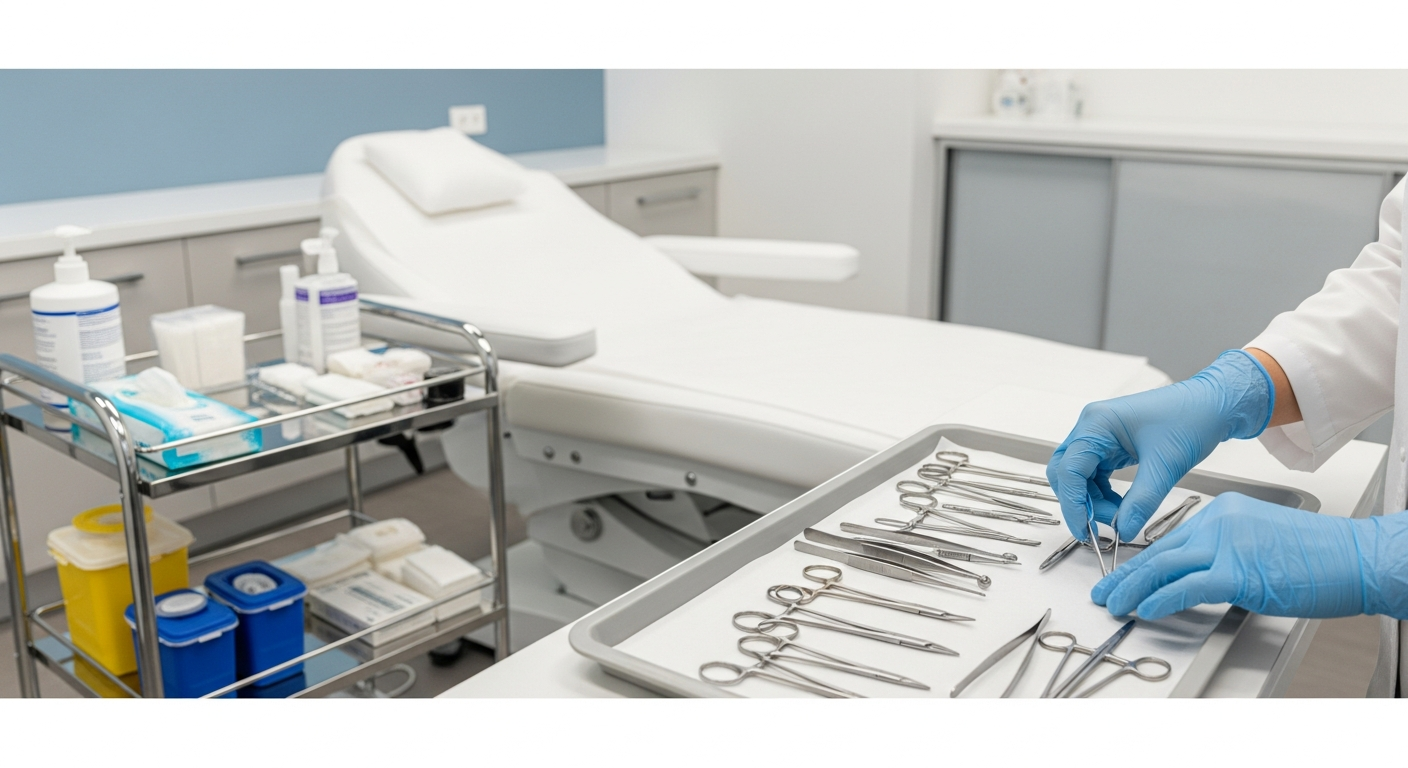
Sterilization Standards for Aesthetic Tools
Any instrument that penetrates the skin or contacts mucosal membranes requires professional sterilization.
Med spas typically utilize autoclaves that use saturated steam under pressure to kill all microbial life.
Routine spore testing is mandatory to verify that the sterilization equipment is functioning correctly.
Managing Reusable vs. Single-Use Items
Strict differentiation between multi-use tools and single-use disposables is critical for safety.
Single-use items, such as lancets or syringe tips, must be discarded in biohazard sharps containers.
Reusable tools must be pre-cleaned, dried, and packaged before entering the sterilization cycle.
Preventing Cross-Contamination in Clinical Settings
Cross-contamination is prevented by establishing clear “clean” and “dirty” zones within the med spa.
Supplies should be stored in closed cabinets to protect them from airborne particles.
Practitioners must avoid touching common surfaces like phones or doorknobs with contaminated gloves.
Implementing a color-coded cleaning system can further reduce the risk of environmental contamination.
Regular audits of sanitation logs ensure that all team members adhere to safety benchmarks.
Consistent safety processes protect the reputation of the med spa and the health of the community.