Med Spa Infection Control and Clinical Safety Standards
Med spa infection control involves the systematic implementation of sanitation, disinfection, and sterilization protocols designed to prevent cross-contamination within aesthetic clinics. These OSHA-aligned practices include rigorous treatment room cleaning, proper sharps handling, and the use of EPA-registered disinfectants to maintain a sterile environment during non-invasive cosmetic procedures.
Core Principles of Med Spa Clinical Safety
Standard precautions serve as the foundation for every med spa procedure. Practitioners must treat all biological materials as potentially infectious to ensure provider and client safety.
Hand hygiene is the most critical step in preventing the spread of pathogens. Staff must wash hands before donning gloves and immediately after removal between every client session.
Consistent use of barriers protects high-touch surfaces from contamination. This includes using disposable covers on treatment tables, equipment screens, and product containers during procedures.
Treatment Room Disinfection Systems
Effective med spa infection control requires a two-step approach to surface management. Surfaces must be physically cleaned to remove debris before the application of any disinfectant.
Clinical surfaces require EPA-registered, hospital-grade disinfectants with specific contact times. Practitioners must ensure surfaces remain wet for the full duration specified by the manufacturer.
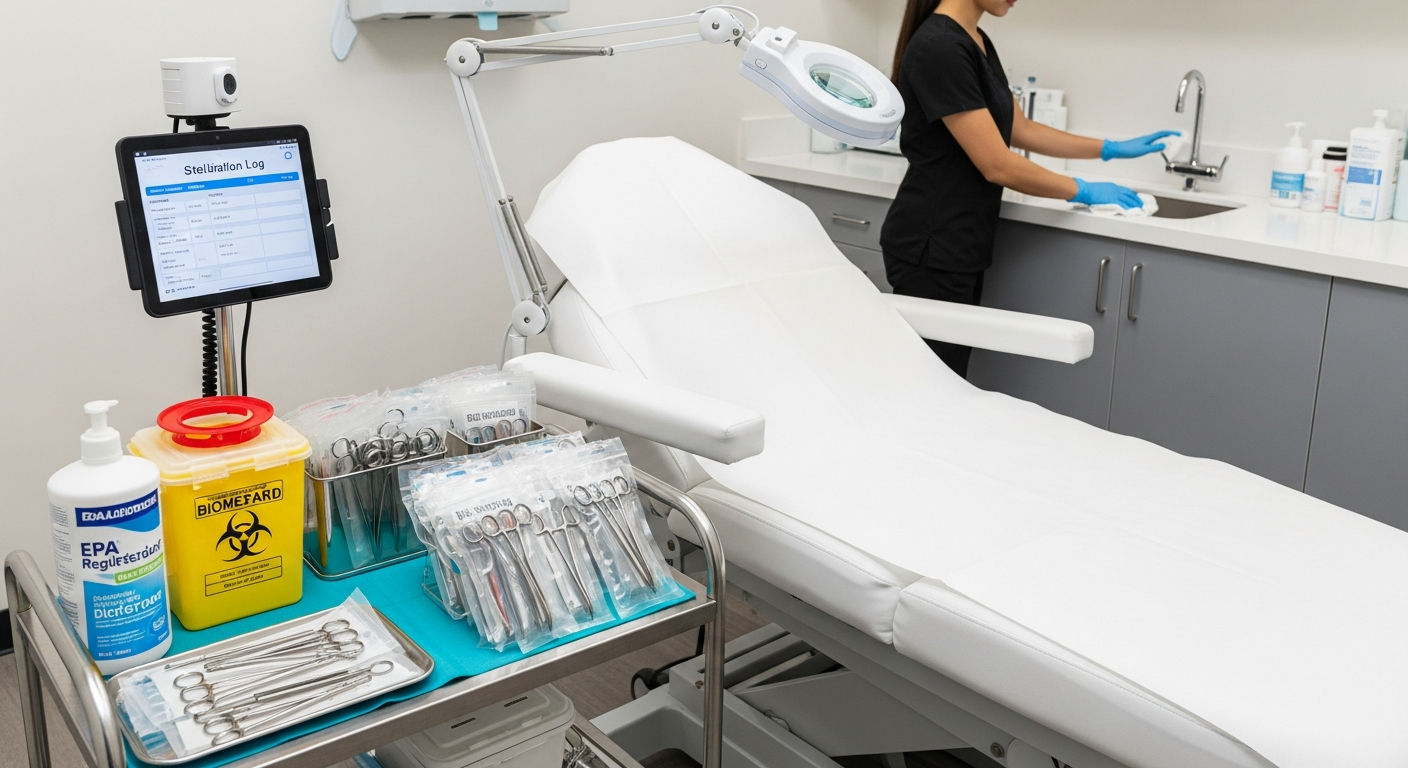
High-touch zones include treatment beds, stools, counter surfaces, and magnifying lamps. These areas must be disinfected between every client without exception to prevent cross-contamination.
Instrument Sterilization Protocols
Any reusable tool that penetrates the skin or contacts mucosal membranes requires professional sterilization. Med spa facilities should utilize medical-grade autoclaves for these specific instruments.
Sterilization logs must be maintained to document every cycle, including temperature, pressure, and duration. This documentation provides a critical audit trail for compliance managers.
Weekly spore testing is necessary to verify that sterilization equipment is functioning correctly. Third-party laboratory verification ensures that the heat and pressure are effectively killing all microbial life.
Personal Protective Equipment (PPE) Standards
PPE acts as a vital barrier between the practitioner and potential contaminants. Med spa staff must select equipment based on the specific risks associated with each treatment type.
Single-use medical gloves must be changed between every client and whenever they become compromised. Gloves should never be washed or reused in a clinical setting.
Fluid-resistant masks and eye protection are essential when performing treatments that may involve splashes or aerosols. This protects the mucous membranes of the practitioner from exposure.
OSHA Compliance and Biohazard Management
Proper disposal of clinical waste is a legal requirement under OSHA standards. Med spa facilities must clearly separate general waste from regulated biohazardous materials.
Sharps containers must be placed within reach of the treatment area. All needles, lancets, and syringes must be discarded immediately into puncture-resistant, leak-proof containers.
Biohazard bags should be used for any materials saturated with blood or other potentially infectious materials. These bags must be stored in a secure area until collected by a licensed disposal service.
Staff Training and Safety Documentation
Continuous education ensures that all team members understand current infection control mandates. Annual training updates should cover bloodborne pathogens and chemical safety protocols.
Safety Data Sheets (SDS) for all clinical cleaning agents must be readily accessible to all staff. These documents outline the necessary first-aid and handling procedures for chemical exposures.
Regular safety audits help clinical supervisors identify gaps in sanitation routines. Establishing a culture of accountability ensures that infection control remains a top priority for the facility.