Med Spa Infection Control and Clinical Safety Protocols
Med spa infection control refers to the systematic application of sanitation and sterilization standards designed to eliminate pathogens in aesthetic treatment environments. These protocols prioritize OSHA-aligned practices, including high-level disinfection of surfaces and the sterilization of reusable tools to prevent cross-contamination and ensure a clinically safe environment for clients and providers.
Regulatory Framework for Med Spa Safety
Operating a safe aesthetic facility requires strict adherence to federal and state safety guidelines. Compliance managers must implement Standard Precautions, treating all body fluids as potentially infectious.
OSHA-aligned practices form the foundation of these systems, focusing on bloodborne pathogen training and hazard communication. Maintaining a Safety Data Sheet (SDS) binder is mandatory for all clinical disinfectants.
Regular safety audits ensure that every team member follows established hygiene cycles. Consistency in these protocols minimizes legal risks and protects the reputation of the med spa.
Multi-Level Sanitation and Disinfection Systems
High-Touch Surface Disinfection
Treatment room surfaces, such as aesthetic beds, counters, and equipment trolleys, require disinfection between every client. Use EPA-registered disinfectants that are specifically rated for clinical environments.
Contact time is the most critical factor in surface disinfection. Ensure the surface remains visibly wet for the full duration specified by the manufacturer to effectively neutralize pathogens.
Environment Maintenance and Air Quality
Infection control extends to the physical environment of the med spa. Frequent cleaning of ventilation grates and high-traffic flooring prevents the accumulation of dust and biological debris.
Implementing a daily terminal cleaning checklist ensures that no area is overlooked. Focus on shared spaces like consultation rooms and post-treatment recovery areas.
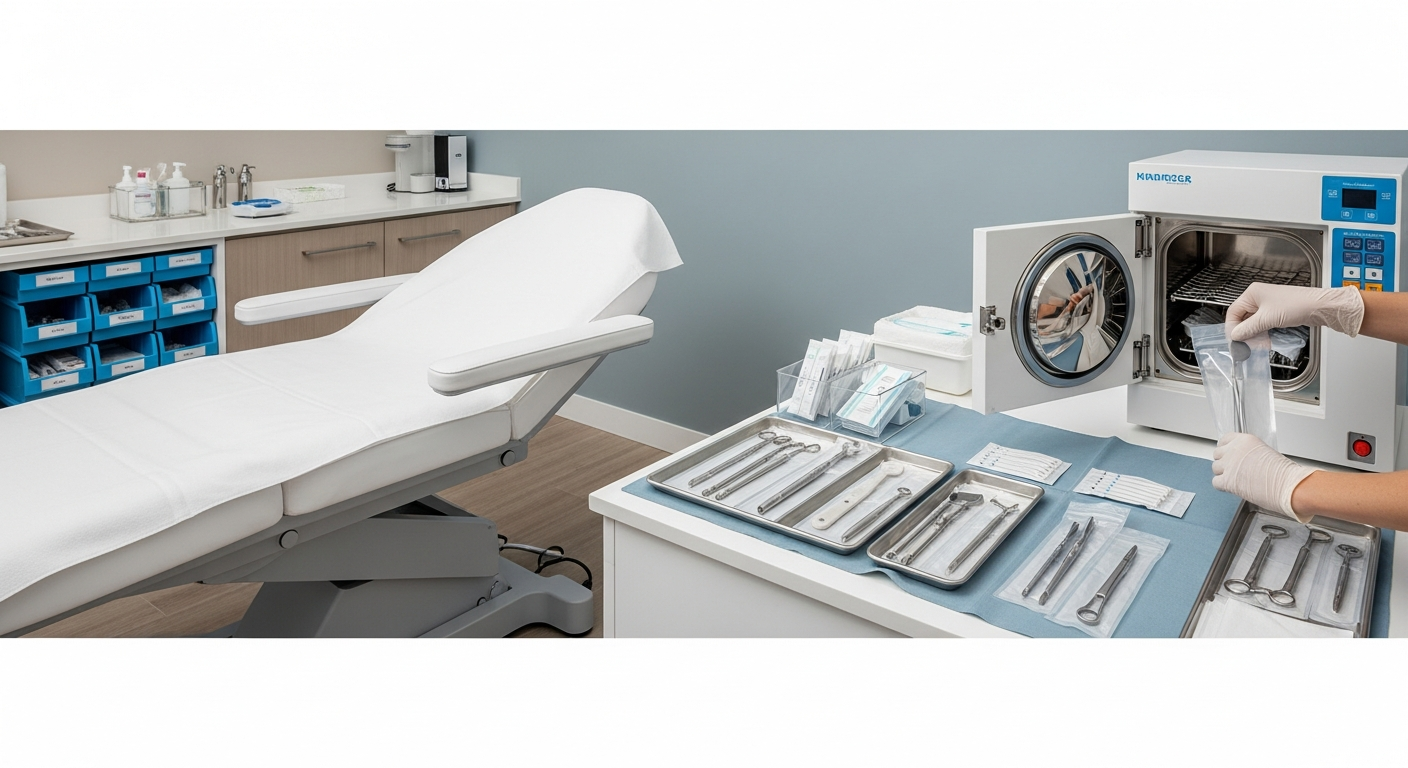
Sterilization Protocols for Aesthetic Tools
Critical vs. Semi-Critical Instruments
Instruments that penetrate the skin are considered critical and must undergo full sterilization. Semi-critical items that touch intact skin require high-level disinfection or single-use disposal.
Using single-use, disposable tools is often the safest method for preventing cross-contamination. When reusable tools are necessary, they must be processed through a validated sterilization system.
Autoclave Operation and Monitoring
Steam sterilization via an autoclave is the industry standard for reusable aesthetic instruments. Tools must be cleaned of all visible debris before being placed in sterilization pouches.
Clinical supervisors must perform weekly biological monitoring, such as spore testing, to verify autoclave efficacy. Maintain a detailed log of every sterilization cycle for compliance records.
Cross-Contamination Prevention Strategies
Personal Protective Equipment (PPE)
Correct PPE usage acts as a primary barrier between the provider and potential contaminants. This includes medical-grade gloves, face masks, and protective eyewear during specific aesthetic procedures.
Gloves must be changed immediately if they become compromised or after every client interaction. PPE should never be worn outside the designated treatment zone to prevent spreading contaminants.
Hand Hygiene Excellence
Rigorous hand hygiene is the single most effective way to prevent the spread of infections. Providers must wash hands with antimicrobial soap before and after every client contact.
Alcohol-based hand rubs are effective for intermediate sanitization but do not replace traditional washing when hands are visibly soiled. Training should emphasize the Five Moments for Hand Hygiene.
Treatment Room Turnover Procedures
Efficient room turnover relies on a systematic checklist to ensure clinical safety. Remove all used linens, disposables, and sharps immediately after the treatment is completed.
All reusable equipment must be wiped down with intermediate-level disinfectants. Reset the room with fresh barrier films on high-touch digital screens and equipment handles.
A standardized turnover process prevents errors and ensures the next client enters a pristine environment. Documented turnover logs help clinical supervisors monitor staff compliance with safety tasks.